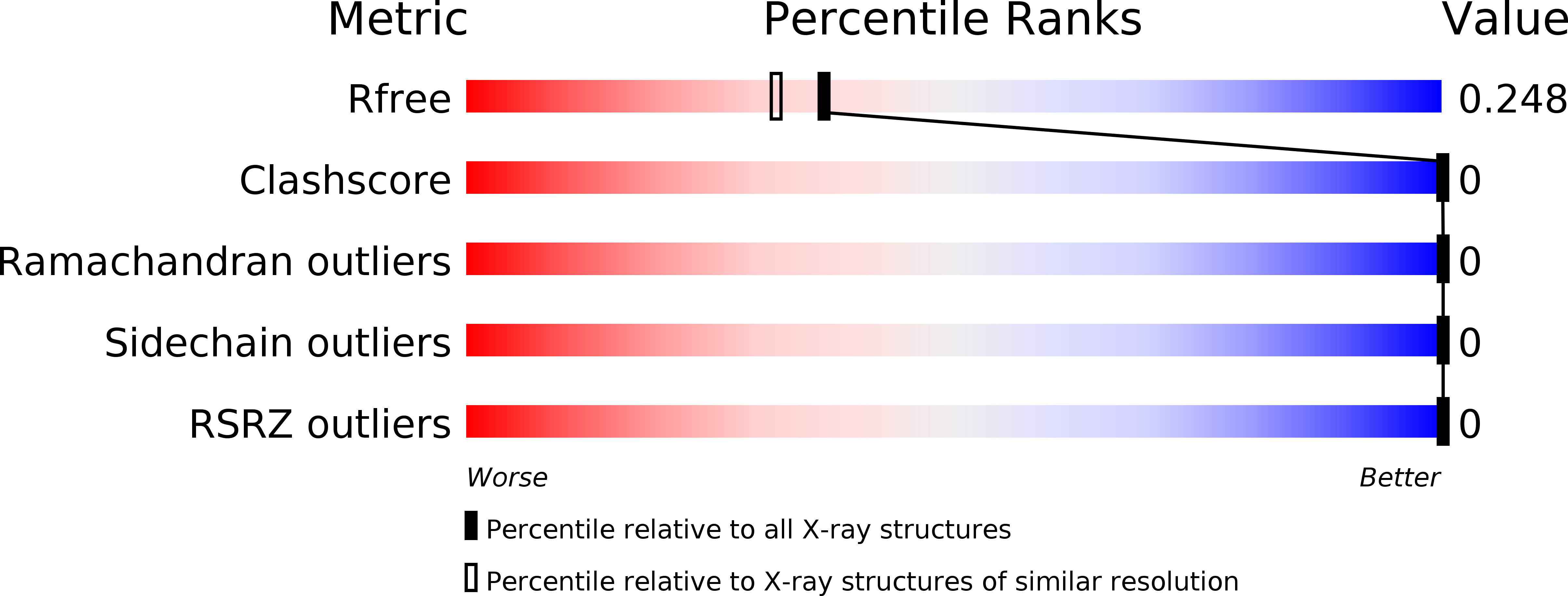
X-ray Crystallographic Structure of a Compact Dodecamer from a Peptide Derived from A beta 16-36.
Salveson, P.J., Spencer, R.K., Kreutzer, A.G., Nowick, J.S.(2017) Org Lett 19: 3462-3465
- PubMed: 28683555
- DOI: https://doi.org/10.1021/acs.orglett.7b01445
- Primary Citation of Related Structures:
5V63, 5V64, 5V65 - PubMed Abstract:
The assembly of the β-amyloid peptide, Aβ, into soluble oligomers is associated with neurodegeneration in Alzheimer's disease. The Aβ oligomers are thought to be composed of β-hairpins. Here, the effect of shifting the residue pairing of the β-hairpins on the structures of the oligomers that form is explored through X-ray crystallography. Three residue pairings were investigated using constrained macrocyclic β-hairpins in which Aβ 30-36 is juxtaposed with Aβ 17-23 , Aβ 16-22 , and Aβ 15-21 . The Aβ 16-22 -Aβ 30-36 pairing forms a compact ball-shaped dodecamer composed of fused triangular trimers. This dodecamer may help explain the structures of the trimers and dodecamers formed by full-length Aβ.
Organizational Affiliation:
Department of Chemistry, University of California, Irvine , Irvine, California 92697-2025, United States.