Crystal Structure of Escherichia coli GroEL with Magnesium Ions and a Phosphorylated Serine Residue
Guo, Y., Zhang, L., Zheng, H., Li, J., Han, Q.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Chaperonin GroEL | 548 | Escherichia coli BL21(DE3) | Mutation(s): 0 EC: 5.6.1.7 | 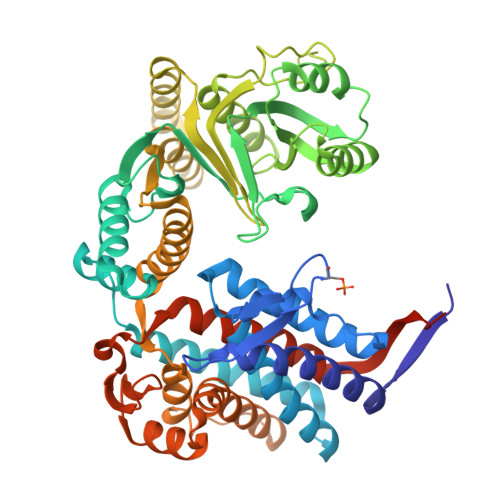 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0A6F5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Chaperonin GroEL | 548 | Escherichia coli BL21(DE3) | Mutation(s): 0 EC: 5.6.1.7 | 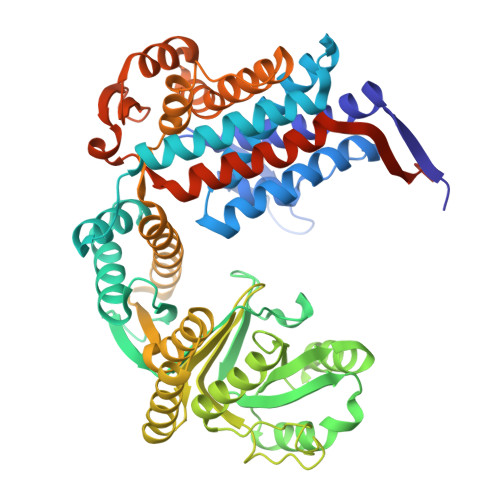 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0A6F5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| TAM Download:Ideal Coordinates CCD File | KB [auth I], PB [auth J], YA [auth G] | TRIS(HYDROXYETHYL)AMINOMETHANE C7 H17 N O3 GKODZWOPPOTFGA-UHFFFAOYSA-N |  | ||
| HEZ Download:Ideal Coordinates CCD File | AC [auth N] BA [auth C] CA [auth C] FB [auth H] GB [auth H] | HEXANE-1,6-DIOL C6 H14 O2 XXMIOPMDWAUFGU-UHFFFAOYSA-N |  | ||
| BME Download:Ideal Coordinates CCD File | AA [auth B] BB [auth G] CB [auth G] CC [auth N] DA [auth C] | BETA-MERCAPTOETHANOL C2 H6 O S DGVVWUTYPXICAM-UHFFFAOYSA-N |  | ||
| MG (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | AB [auth G] BC [auth N] HB [auth H] JA [auth D] PA [auth E] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| SEP Query on SEP | A, B, E, F, H A, B, E, F, H, J, M | L-PEPTIDE LINKING | C3 H8 N O6 P |  | SER |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 135.835 | α = 90 |
| b = 262.165 | β = 90 |
| c = 284.411 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| SCALEPACK | data scaling |
| HKL-2000 | data reduction |
| MOLREP | phasing |
| PDB_EXTRACT | data extraction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of Neurological Disorders and Stroke (NIH/NINDS) | United States | NS062836 |
| National Natural Science Foundation of China (NSFC) | China | 31860702 |