Computationally designed tunable C2 symmetric tandem repeat homodimer, D_3_633_8x bound to peptide
Kennedy, M.A., Stoddard, B.L., Hicks, D.R.To be published.
Experimental Data Snapshot
Starting Model: in silico
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| D_3_633_8x peptide bound | 234 | synthetic construct | Mutation(s): 0 | 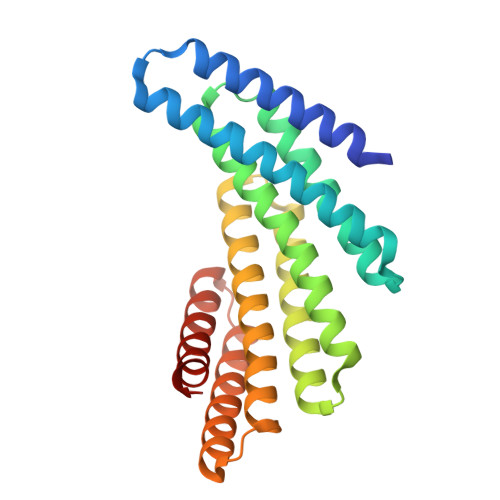 | |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| MC1 | C [auth D] | 8 | synthetic construct | Mutation(s): 0 | 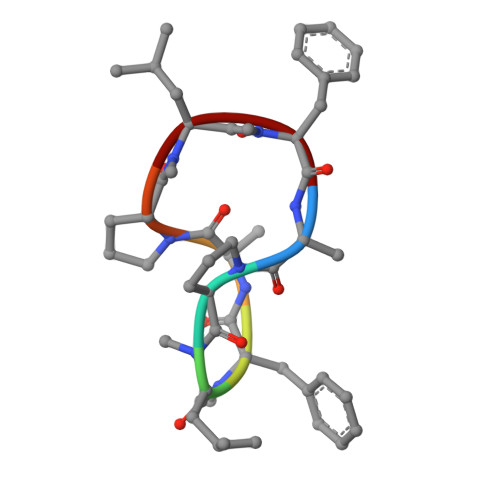 |
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SO4 Download:Ideal Coordinates CCD File | E [auth B], F [auth B], G [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| POL Download:Ideal Coordinates CCD File | D [auth A], H [auth B], I [auth B] | N-PROPANOL C3 H8 O BDERNNFJNOPAEC-UHFFFAOYSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| DAL Query on DAL | C [auth D] | D-PEPTIDE LINKING | C3 H7 N O2 |  | -- |
| DPR Query on DPR | C [auth D] | D-PEPTIDE LINKING | C5 H9 N O2 |  | -- |
| MEA Query on MEA | C [auth D] | L-PEPTIDE LINKING | C10 H13 N O2 |  | PHE |
| MLE Query on MLE | C [auth D] | L-PEPTIDE LINKING | C7 H15 N O2 |  | LEU |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 54.717 | α = 90 |
| b = 54.562 | β = 96.11 |
| c = 80.478 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data scaling |
| PHASER | phasing |
| HKL-2000 | data collection |
| HKL-2000 | data reduction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Science Foundation (NSF, United States) | United States | DGE-1762114 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | R01GM115545 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | R01GM139752 |
| National Institutes of Health/National Institute on Aging (NIH/NIA) | United States | R01AG063845 |
| National Institutes of Health/National Institute on Aging (NIH/NIA) | United States | U19AG065156 |