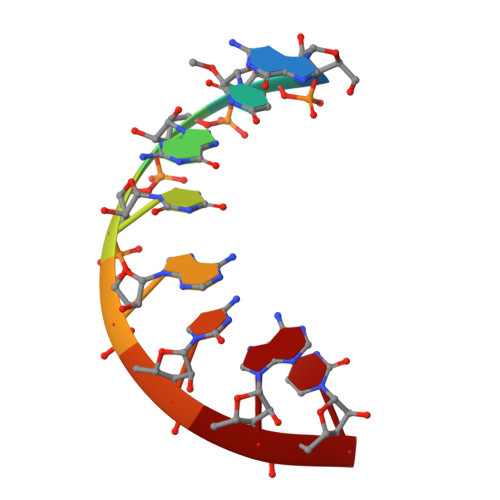
Derivatization of Mirror-Image l-Nucleic Acids with 2'-OMe Modification for Thermal and Structural Stabilization.
Dantsu, Y., Zhang, W.(2023) Chembiochem 24: e202200764-e202200764
- PubMed: 36892526 Search on PubMed
- DOI: https://doi.org/10.1002/cbic.202200764
- Primary Citation Related Structures:
8F24, 8F27 - PubMed Abstract:
To further expand the functionality and enhance the stability of mirror-image nucleic acids as advanced agents for basic research and therapeutic design, we have synthesized 2'-deoxy-2'-methoxy-l-uridine phosphoramidite and incorporated it into l-DNA and l-RNA by solid-phase synthesis quantitatively. We found that the thermostability of l-nucleic acids is dramatically improved after introducing the modifications. Moreover, we successfully crystallized both l-DNA and l-RNA duplexes containing the 2'-OMe modifications and sharing identical sequences. Crystal structure determination and analysis revealed the overall structures of the mirror-image nucleic acids, and for the first time it was possible to interpret the structural deviations caused by 2'-OMe and 2'-OH groups in the oligonucleotides, which are very similar. This novel chemical nucleic acid modification has the potential to be used to design nucleic acid-based therapeutics and materials in the future.
- Department of Biochemistry and Molecular Biology, Indiana University School of Medicine, 635 Barnhill Drive, Indianapolis, IN, 46202, USA.
Organizational Affiliation: