The Corramycin phosphotransferase in complex with Corramycin
Adam, S., Mueller, R., Koehnke, J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Corramycin phosphotransferase | 366 | Corallococcus coralloides | Mutation(s): 0 | 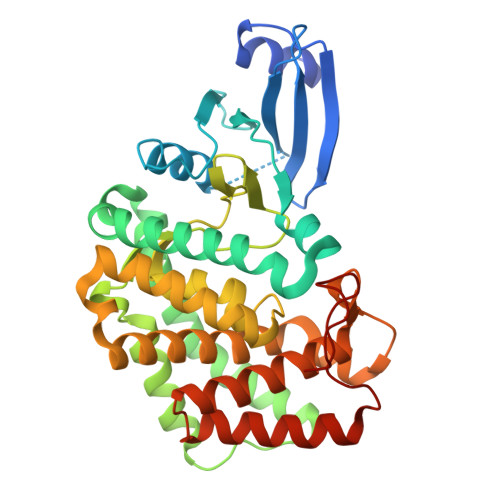 | |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Corramycin | 10 | Corallococcus coralloides | Mutation(s): 0 | 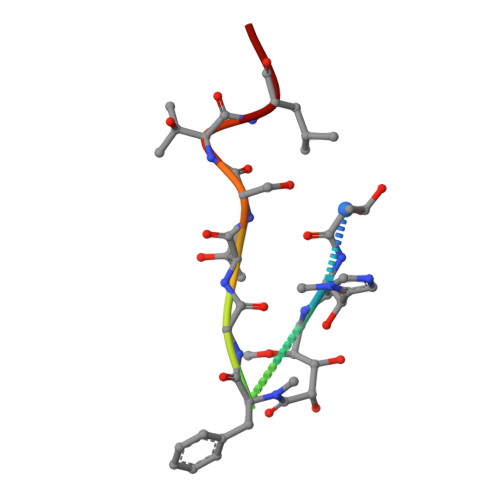 | |
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| GOL Download:Ideal Coordinates CCD File | C [auth A], D [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Modified Residues 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| HVA Query on HVA | B | L-PEPTIDE LINKING | C5 H11 N O3 |  | VAL |
| MEA Query on MEA | B | L-PEPTIDE LINKING | C10 H13 N O2 |  | PHE |
| OGC Query on OGC | B | L-PEPTIDE LINKING | C7 H11 N3 O3 |  | HIS |
| Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_002426 Query on PRD_002426 | B | Corramycin | Peptide-like / Antibiotic, Antimicrobial |  | |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 42.964 | α = 90 |
| b = 67.116 | β = 93.33 |
| c = 75.553 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Coot | model building |
| XDS | data reduction |
| SCALA | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Helmholtz Association | Germany | -- |