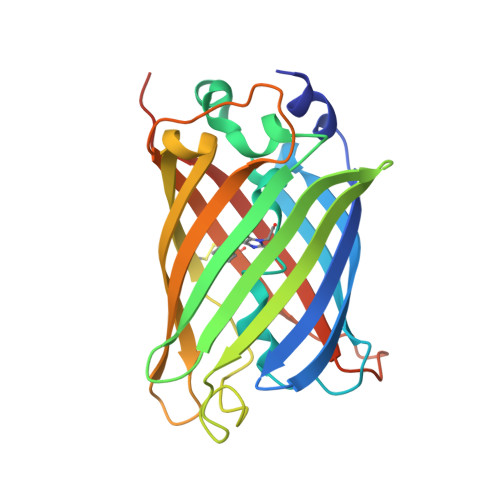
Crystal Structure of Green Fluorescent Protein Clover and Design of Clover-Based Redox Sensors.
Campbell, B.C., Petsko, G.A., Liu, C.F.(2018) Structure 26: 225-237.e3
- PubMed: 29307487 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2017.12.006
- Primary Citation Related Structures:
5WJ2, 5WJ3, 5WJ4 - PubMed Abstract:
We have determined the crystal structure of Clover, one of the brightest fluorescent proteins, and found that its T203H/S65G mutations relative to wild-type GFP lock the critical E222 side chain in a fixed configuration that mimics the major conformer of that in EGFP. The resulting equilibrium shift to the predominantly deprotonated chromophore increases the extinction coefficient (EC), opposes photoactivation, and is responsible for the bathochromic shift. Clover's brightness can further be attributed to a π-π stacking interaction between H203 and the chromophore. Consistent with these observations, the Clover G65S mutant reversed the equilibrium shift, dramatically decreased the EC, and made Clover photoactivatable under conditions that activated photoactivatable GFP. Using the Clover structure, we rationally engineered a non-photoactivatable redox sensor, roClover1, and determined its structure as well as that of its parental template, roClover0.1. These high-resolution structures provide deeper insights into structure-function relationships in GFPs and may aid the development of excitation-improved ratiometric biosensors.
- Helen and Robert Appel Alzheimer's Disease Research Institute and Feil Family Brain and Mind Research Institute, Weill Cornell Medicine, New York, NY 10021, USA.
Organizational Affiliation: