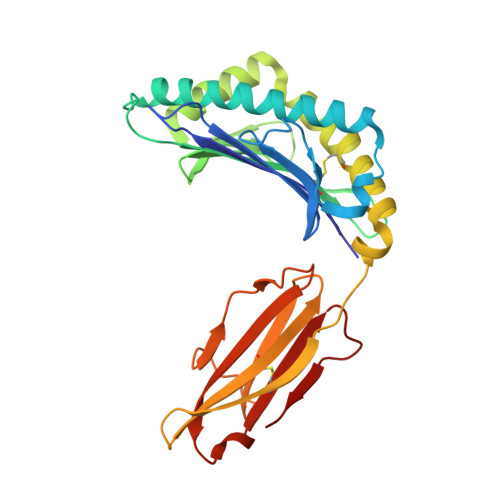
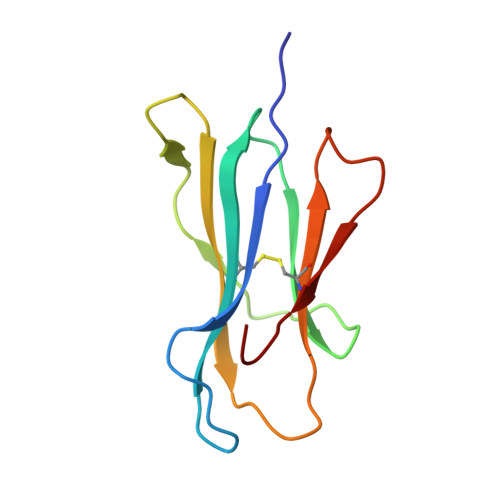
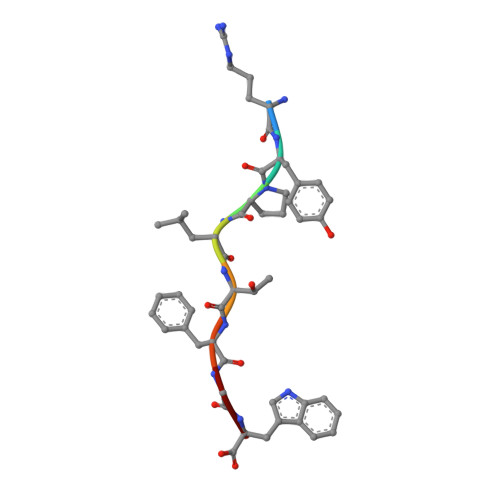
Effects of a Single Escape Mutation on T Cell and HIV-1 Co-adaptation.
Sun, X., Shi, Y., Akahoshi, T., Fujiwara, M., Gatanaga, H., Schonbach, C., Kuse, N., Appay, V., Gao, G.F., Oka, S., Takiguchi, M.(2016) Cell Rep 15: 2279-2291
- PubMed: 27239036 Search on PubMed
- DOI: https://doi.org/10.1016/j.celrep.2016.05.017
- Primary Citation Related Structures:
5HGA, 5HGB, 5HGD, 5HGH - PubMed Abstract:
The mechanistic basis for the progressive accumulation of Y(135)F Nef mutant viruses in the HIV-1-infected population remains poorly understood. Y(135)F viruses carry the 2F mutation within RW8 and RF10, which are two HLA-A(∗)24:02-restricted superimposed Nef epitopes recognized by distinct and adaptable CD8(+) T cell responses. We combined comprehensive analysis of the T cell receptor repertoire and cross-reactive potential of wild-type or 2F RW8- and RF10-specific CD8(+) T cells with peptide-MHC complex stability and crystal structure studies. We find that, by affecting direct and water-mediated hydrogen bond networks within the peptide-MHC complex, the 2F mutation reduces both TCR and HLA binding. This suggests an advantage underlying the evolution of the 2F variant with decreased CD8(+) T cell efficacy. Our study provides a refined understanding of HIV-1 and CD8(+) T cell co-adaptation at the population level.
- Center for AIDS Research, Kumamoto University, Kumamoto 860-0811, Japan.
Organizational Affiliation: