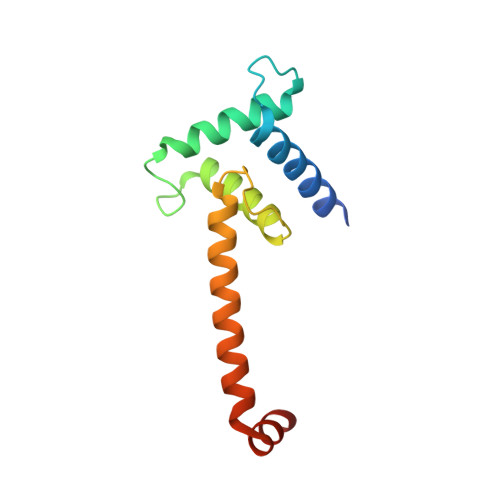
Structural insights into interactions of C/EBP transcriptional activators with the Taz2 domain of p300.
Bhaumik, P., Davis, J., Tropea, J.E., Cherry, S., Johnson, P.F., Miller, M.(2014) Acta Crystallogr D Biol Crystallogr 70: 1914-1921
- PubMed: 25004968 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S1399004714009262
- Primary Citation Related Structures:
3T92 - PubMed Abstract:
Members of the C/EBP family of transcription factors bind to the Taz2 domain of p300/CBP and mediate its phosphorylation through the recruitment of specific kinases. Short sequence motifs termed homology boxes A and B, which comprise their minimal transactivation domains (TADs), are conserved between C/EBP activators and are necessary for specific p300/CBP binding. A possible mode of interaction between C/EBP TADs and the p300 Taz2 domain was implied by the crystal structure of a chimeric protein composed of residues 1723-1818 of p300 Taz2 and residues 37-61 of C/EBPℇ. The segment corresponding to the C/EBPℇ TAD forms two orthogonally disposed helices connected by a short linker and interacts with the core structure of Taz2 from a symmetry-related molecule. It is proposed that other members of the C/EBP family interact with the Taz2 domain in the same manner. The position of the C/EBPℇ peptide on the Taz2 protein interaction surface suggests that the N-termini of C/EBP proteins are unbound in the C/EBP-p300 Taz2 complex. This observation is in agreement with the known location of the docking site of protein kinase HIPK2 in the C/EBPβ N-terminus, which associates with the C/EBPβ-p300 complex.
- Protein Structure Section, Macromolecular Crystallography Laboratory, National Cancer Institute at Frederick, Frederick, MD 21702, USA.
Organizational Affiliation: