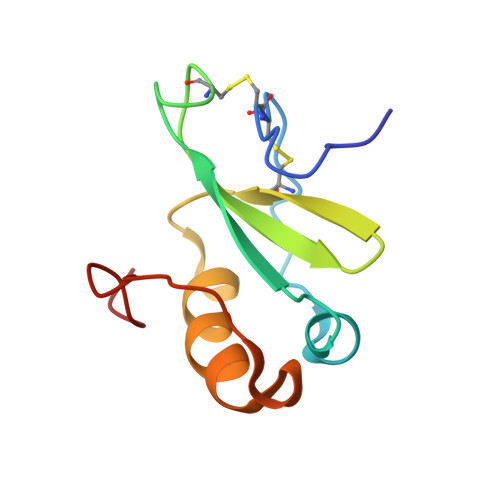
NMR analysis of the structure, dynamics, and unique oligomerization properties of the chemokine CCL27.
Jansma, A.L., Kirkpatrick, J.P., Hsu, A.R., Handel, T.M., Nietlispach, D.(2010) J Biological Chem 285: 14424-14437
- PubMed: 20200157 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M109.091108
- Primary Citation Related Structures:
2KUM - PubMed Abstract:
Chemokines have two essential interactions in vivo, with G protein-coupled receptors, which activate intracellular signaling pathways, and with glycosaminoglycans (GAGs), which are involved in cell surface localization and transport. Although it has been shown that chemokines bind and activate their respective G protein-coupled receptors as monomers, many chemokines oligomerize upon GAG binding, and the ability to oligomerize and bind GAGs is required for in vivo function. In this study, we investigated the structure, dynamics, and oligomerization behavior of cutaneous T-cell-attracting chemokine (CTACK, also known as CCL27) by NMR. (15)N relaxation and translational self-diffusion rates indicate that CCL27 oligomerizes, but in contrast to many other chemokines that form relatively discrete oligomers, CCL27 transitions between monomer, dimer, and tetramer species over a relatively narrow concentration range. A three-dimensional structure determination was pursued under conditions where CCL27 is primarily dimeric, revealing the standard motif for a chemokine monomer. Analysis of chemical shift perturbations of (1)H-(15)N HSQC spectra, relaxation-dispersion experiments, and filtered nuclear Overhauser effects suggest that CCL27 does not adopt a discrete CXC or CC dimer motif. Instead, CCL27 has uncommon oligomerization behavior, where several equilibria involving relatively low affinity interactions between different interfaces seem to be simultaneously at work. However, interaction with heparin avidly promotes oligomerization under conditions where CCL27 is monomeric by itself. We hypothesize that the plasticity in the oligomerization state may enable CCL27 to adopt different oligomeric structures, depending on the nature of the GAG binding partner, thereby providing a mechanism for increased diversity and specificity in GAG-binding and GAG-related functions.
- Skaggs School of Pharmacy and Pharmaceutical Science, University of California, San Diego, La Jolla, CA 92093-0684, USA.
Organizational Affiliation: