The Structure of Tomato Aspermy Virus by X-Ray Crystallography
Lucas, R.W., Larson, S.B., Canady, M.A., McPherson, A.(2002) J Struct Biol 139: 90-102
- PubMed: 12406691
- DOI: https://doi.org/10.1016/s1047-8477(02)00561-0
- Primary Citation of Related Structures:
1LAJ - PubMed Abstract:
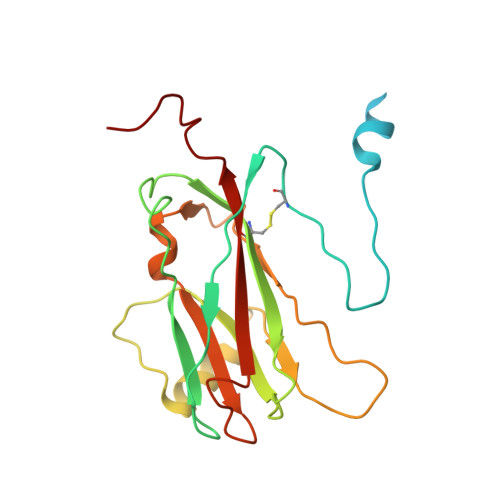
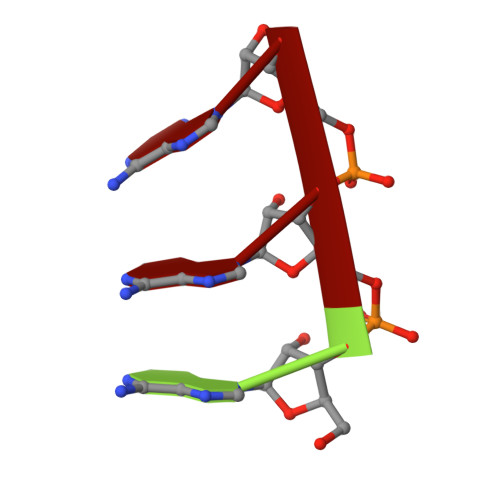
The three-dimensional structure of tomato aspermy virus (TAV) has been solved by X-ray crystallography and refined to an R factor of 0.218 for 3.4-40 A data (effective resolution of 4A). Molecular replacement, using cucumber mosaic virus (Smith et al., 2000), provided phases for the initial maps used for model building. The coat protein of the 280 A diameter virion has the canonical "Swiss roll" beta-barrel topology with a distinctive amino-terminal alpha-helix directed into the interior of the virus where it interacts with encapsidated RNA. The N-terminal helices are joined to the beta-barrels of protein subunits by extended polypeptides of six amino acids, which serve as flexible hinges allowing movement of the helices in response to local RNA distribution. Segments of three nucleotides of partially disordered RNA interact with the capsid, primarily through arginine residues, at interfaces between A and B subunits. Side chains of cys64 and cys106 form the first disulfide observed in a cucumovirus, including a unique cysteine, 106, in a region otherwise conserved. A positive ion, putatively modeled as a Mg(+)ion, lies on the quasi-threefold axis surrounded by three quasi-symmetric glutamate 175 side chains.
- Department of Molecular Biology and Biochemistry, University of California, Irvine, Irvine, CA 92697-3900, USA.
Organizational Affiliation: