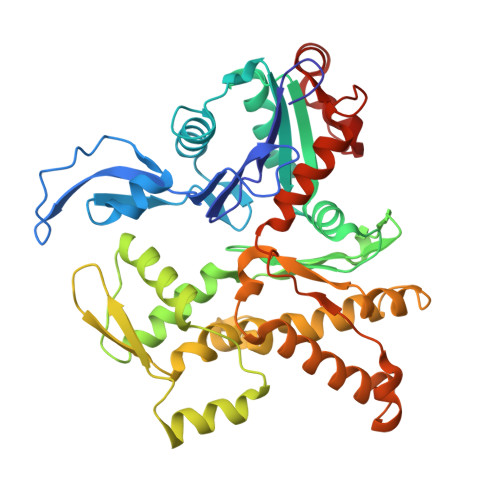
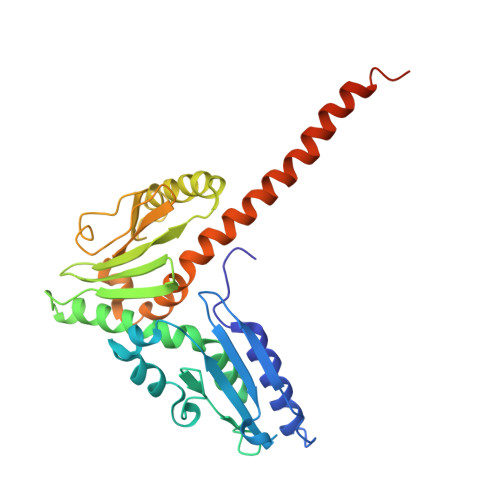
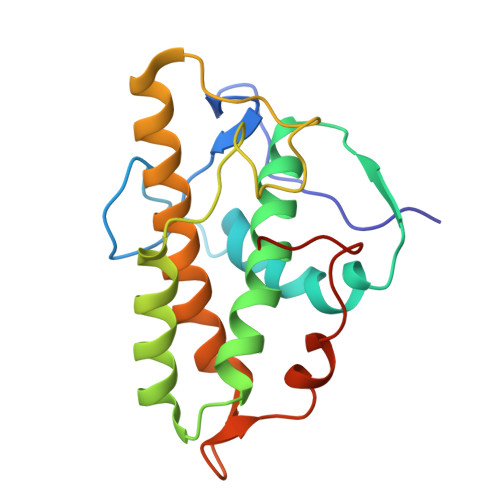
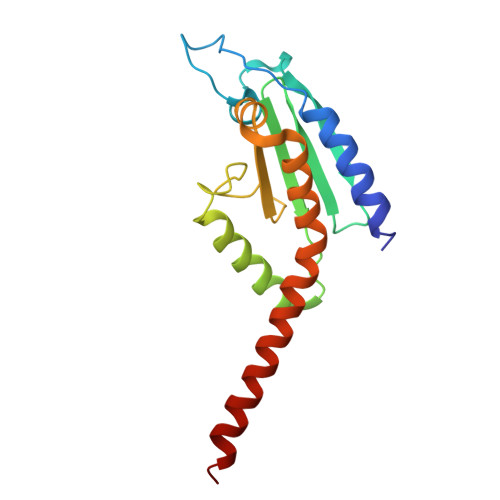
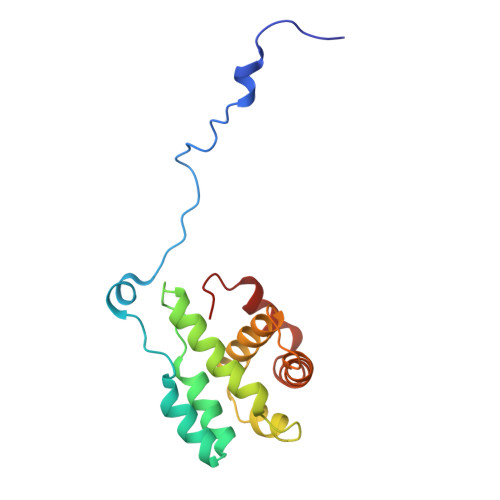
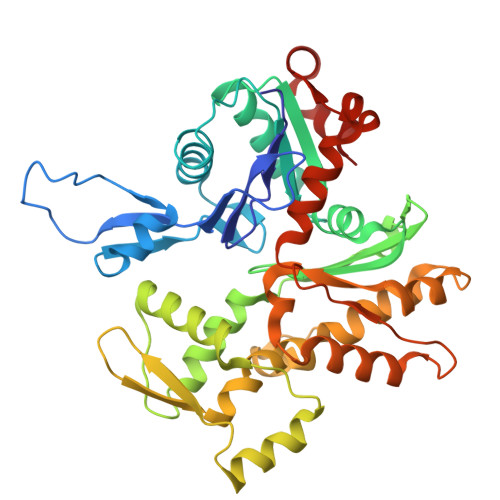
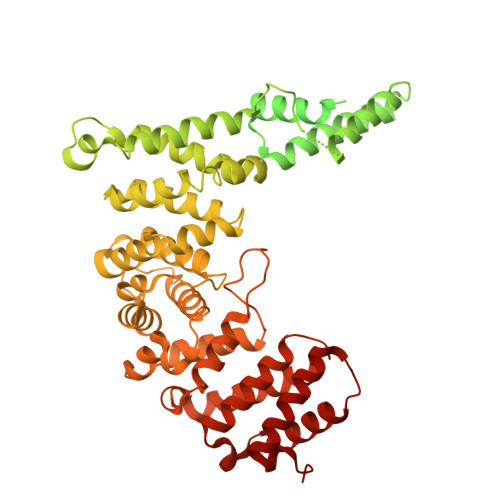
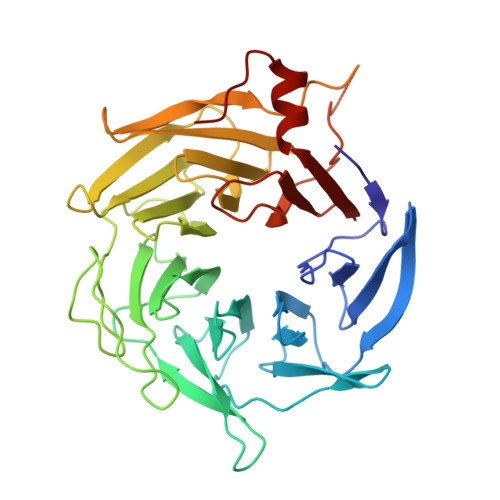
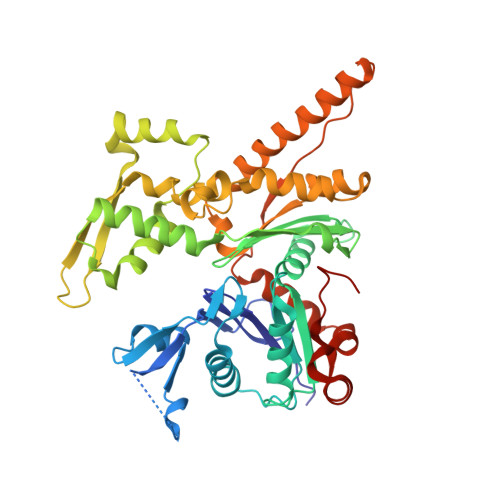
Arp2/3-mediated bidirectional actin assembly by SPIN90 dimers.
Liu, T., Cao, L., Mladenov, M., Romet-Lemonne, G., Way, M., Moores, C.A.(2025) Nat Struct Mol Biol 32: 2262-2271
- PubMed: 40954369 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-025-01665-8
- Primary Citation Related Structures:
9I2B - PubMed Abstract:
Branched actin networks nucleated by the Arp2/3 complex have critical roles in various cellular processes, from cell migration to intracellular transport. However, when activated by WISH/DIP/SPIN90-family proteins, Arp2/3 nucleates linear actin filaments. Here we found that human SPIN90 is a dimer that can nucleate bidirectional actin filaments. To understand the basis for this, we determined a 3-Å-resolution structure of human SPIN90-Arp2/3 complex nucleating actin filaments. Our structure shows that SPIN90 dimerizes through a three-helix bundle and interacts with two Arp2/3 complexes. Each SPIN90 molecule binds both Arp2/3 complexes to promote their activation. Our analysis demonstrates that single-filament nucleation by Arp2/3 is mechanistically more like branch formation than previously appreciated. The dimerization domain in SPIN90 orthologs is conserved in metazoans, suggesting that this mode of bidirectional nucleation is a common strategy to generate antiparallel actin filaments.
- Institute of Structural and Molecular Biology, Birkbeck College, London, UK.
Organizational Affiliation: