Photosensitizer Repositioning Affords an Enantiocomplementary Enzyme for [2 + 2]-Cycloadditions.
Sun, C., Kohn, A.R., Smithson, R., Hardy, F.J., Trimble, J.S., Cao, Y., Johannissen, L.O., Hay, S., Crawshaw, R., Green, A.P.(2025) Angew Chem Int Ed Engl 64: e202503576-e202503576
- PubMed: 40576080 Search on PubMed
- DOI: https://doi.org/10.1002/anie.202503576
- Primary Citation Related Structures:
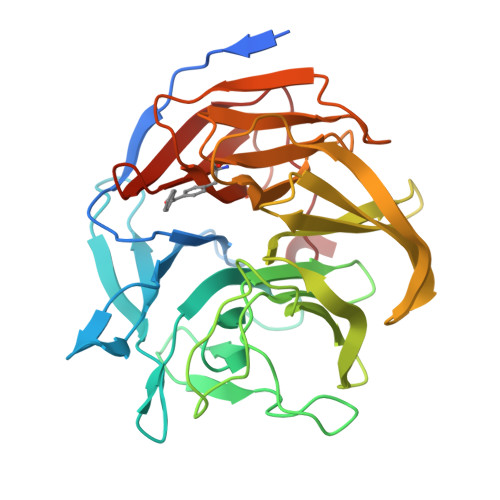
9ENO - PubMed Abstract:
The combination of genetic code expansion and directed evolution has recently given rise to enantioselective photoenzymes for [2 + 2]-cycloadditions of quinolone and indole derivatives. However, the enzymes reported to date only allow access to one enantiomeric series of the strained cyclobutane products. Here, guided by a crystal structure of our previously engineered enzyme EnT1.3, we show how judicious repositioning of the genetically programmed benzophenone photosensitizer affords an enantiocomplementary [2 + 2]-cyclase, CEnT1.0. Following directed evolution, a proficient and oxygen-tolerant photoenzyme (CEnT1.4) emerged that promotes [2 + 2]-cycloadditions of a quinolone derivative with exquisite enantiocontrol (99% e.e.) and substantially enhanced regioselectivity compared with EnT1.3 (r.r. 62:1 vs. 9:1). Structural analysis of CEnT1.4, coupled with molecular dynamic simulations, reveals a well-sculpted active site pocket that pre-organises the substrate for regio- and enantioselective catalysis. This study highlights the versatility offered by genetically programmed (photo)catalytic elements when developing enzymes for altered stereochemical outcomes.
- Department of Chemistry & Manchester Institute of Biotechnology, The University of Manchester, 131 Princess Street, Manchester, M1 7DN, UK.
Organizational Affiliation: