A splendid molecular factory: De- and reconstruction of the mammalian respiratory chain.
Rimle, L., Phillips, B.P., Codo Costa Barra, I.M., Arnold, N., Hennebert, C., Meier, T., von Ballmoos, C.(2025) Proc Natl Acad Sci U S A 122: e2416162122-e2416162122
- PubMed: 40100632 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2416162122
- Primary Citation Related Structures:
8P65 - PubMed Abstract:
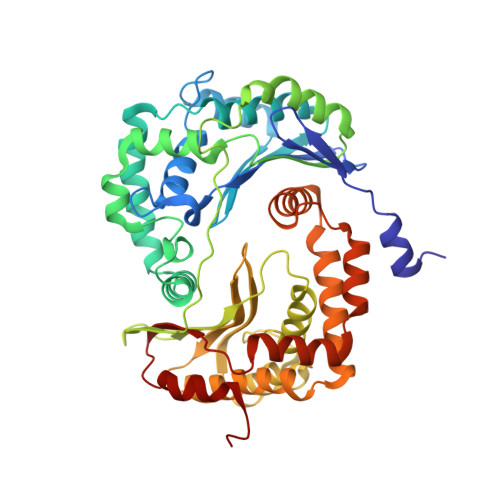
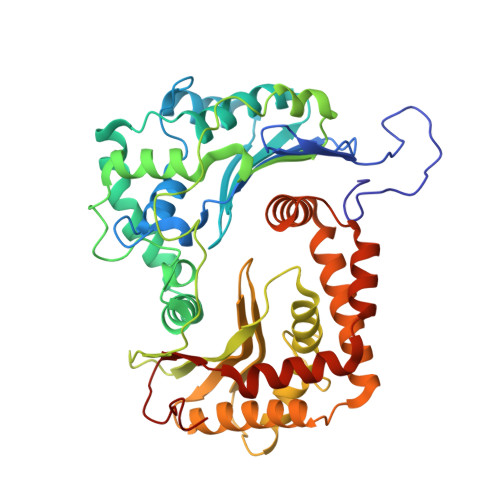
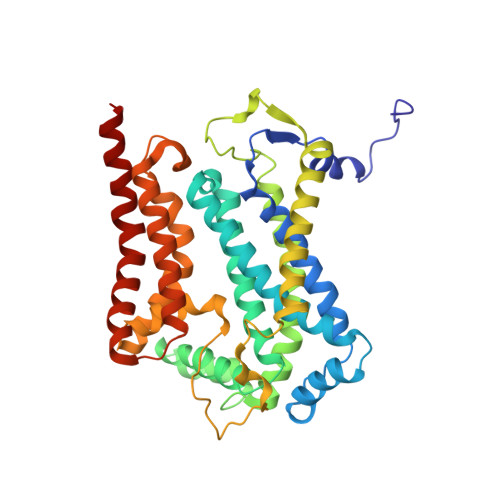
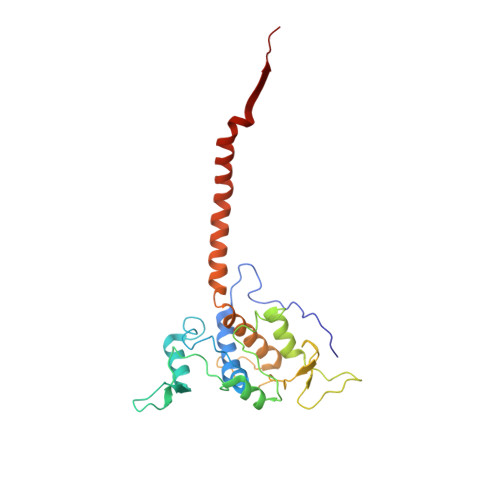
Mitochondrial respiratory complexes I to IV and the F 1 F o -ATP synthase (complex V) are large protein assemblies producing the universal cellular energy currency adenosine triphosphate (ATP). Individual complexes have been extensively studied in vitro, but functional co-reconstitution of several mammalian complexes into proteoliposomes, in particular, the combination of a primary pump with the ATP synthase, is less well understood. Here, we present a generic and scalable strategy to purify mammalian respiratory complexes I, III and the ATP synthase from enriched mitochondria in enzymatically fully active form, and procedures to reassemble the complexes into liposomes. A robust functionality can be shown by in situ monitoring of ATP synthesis rates and by using selected inhibitors of the respiratory chain complexes. By inclusion of cytochrome c oxidase, our procedures allowed us to reconstruct the entire mitochondrial respiratory chain (complexes I, III, IV, and V) in ubiquinone Q 10 containing liposomes, demonstrating oxidative phosphorylation by nicotinamide adenine dinucleotide hydrogen driven ATP synthesis. The system was fully coupled at all levels and was used to probe cardiolipin as an essential component to activate the mammalian respiratory chain. Structural characterization using electron cryomicroscopy allowed us to resolve apo-state complex III and complex V at high and medium resolution, respectively, using in silico particle sorting, confirming the presence of all protein subunits and cofactors in native stoichiometry and conformation. The reported findings will facilitate future endeavors to characterize or modulate these key bioenergetic processes.
- Department of Chemistry, Biochemistry and Pharmaceutical Sciences, University of Bern, Bern 3012, Switzerland.
Organizational Affiliation: