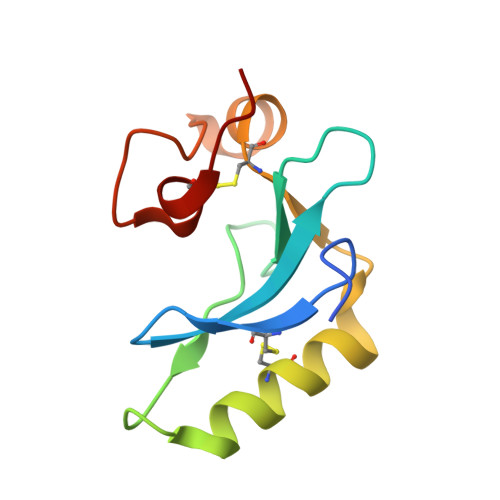
Structural and functional analyses of viral H2 protein of the vaccinia virus entry fusion complex.
Kao, C.F., Liu, C.Y., Hsieh, C.L., Carillo, K.J.D., Tzou, D.L.M., Wang, H.C., Chang, W.(2023) J Virol 97: e0134323-e0134323
- PubMed: 37975688
- DOI: https://doi.org/10.1128/jvi.01343-23
- Primary Citation Related Structures:
8INI - PubMed Abstract:
Vaccinia virus infection requires virus-cell membrane fusion to complete entry during endocytosis; however, it contains a large viral fusion protein complex of 11 viral proteins that share no structure or sequence homology to all the known viral fusion proteins, including type I, II, and III fusion proteins. It is thus very challenging to investigate how the vaccinia fusion complex works to trigger membrane fusion with host cells. In this study, we crystallized the ectodomain of vaccinia H2 protein, one component of the viral fusion complex. Furthermore, we performed a series of mutational, biochemical, and molecular analyses and identified two surface loops containing 170 LGYSG 174 and 125 RRGTGDAW 132 as the A28-binding region. We also showed that residues in the N-terminal helical region (amino acids 51-90) are also important for H2 function.
- Institute of Molecular Biology, Academia Sinica, Taipei, Taiwan.
Organizational Affiliation: