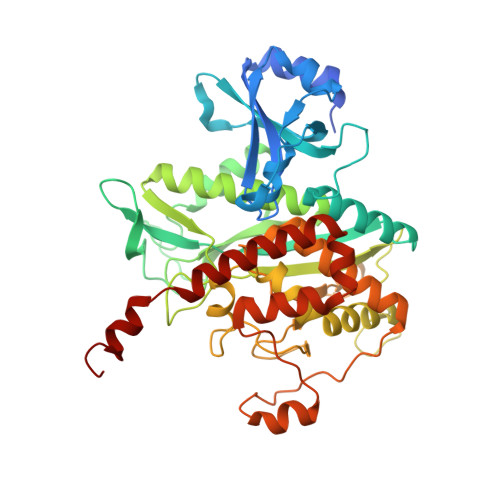
Molecular dissection of the glutamine synthetase-GlnR nitrogen regulatory circuitry in Gram-positive bacteria.
Travis, B.A., Peck, J.V., Salinas, R., Dopkins, B., Lent, N., Nguyen, V.D., Borgnia, M.J., Brennan, R.G., Schumacher, M.A.(2022) Nat Commun 13: 3793-3793
- PubMed: 35778410 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-022-31573-0
- Primary Citation Related Structures:
7TDP, 7TDV, 7TEA, 7TEC, 7TEN, 7TF6, 7TF7, 7TF9, 7TFA, 7TFB, 7TFC, 7TFD, 7TFE - PubMed Abstract:
How bacteria sense and respond to nitrogen levels are central questions in microbial physiology. In Gram-positive bacteria, nitrogen homeostasis is controlled by an operon encoding glutamine synthetase (GS), a dodecameric machine that assimilates ammonium into glutamine, and the GlnR repressor. GlnR detects nitrogen excess indirectly by binding glutamine-feedback-inhibited-GS (FBI-GS), which activates its transcription-repression function. The molecular mechanisms behind this regulatory circuitry, however, are unknown. Here we describe biochemical and structural analyses of GS and FBI-GS-GlnR complexes from pathogenic and non-pathogenic Gram-positive bacteria. The structures show FBI-GS binds the GlnR C-terminal domain within its active-site cavity, juxtaposing two GlnR monomers to form a DNA-binding-competent GlnR dimer. The FBI-GS-GlnR interaction stabilizes the inactive GS conformation. Strikingly, this interaction also favors a remarkable dodecamer to tetradecamer transition in some GS, breaking the paradigm that all bacterial GS are dodecamers. These data thus unveil unique structural mechanisms of transcription and enzymatic regulation.
- Department of Biochemistry, 307 Research Dr., Box 3711, Duke University Medical Center, Durham, NC, 27710, USA.
Organizational Affiliation: