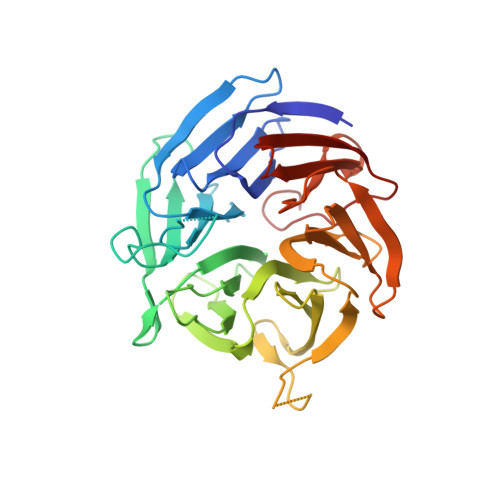
A broad specificity beta-propeller enzyme from Rhodopseudomonas palustris that hydrolyzes many lactones including gamma-valerolactone.
Hall, B.W., Bingman, C.A., Fox, B.G., Noguera, D.R., Donohue, T.J.(2022) J Biological Chem 299: 102782-102782
- PubMed: 36502920 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jbc.2022.102782
- Primary Citation Related Structures:
7RIS, 7RIZ, 8DJF, 8DJZ, 8DK0 - PubMed Abstract:
Lactones are prevalent in biological and industrial settings, yet there is a lack of information regarding enzymes used to metabolize these compounds. One compound, γ-valerolactone (GVL), is used as a solvent to dissolve plant cell walls into sugars and aromatic molecules for subsequent microbial conversion to fuels and chemicals. Despite the promise of GVL as a renewable solvent for biomass deconstruction, residual GVL can be toxic to microbial fermentation. Here, we identified a Ca 2+ -dependent enzyme from Rhodopseudomonas palustris (Rpa3624) and showed that it can hydrolyze aliphatic and aromatic lactones and esters, including GVL. Maximum-likelihood phylogenetic analysis of other related lactonases with experimentally determined substrate preferences shows that Rpa3624 separates by sequence motifs into a subclade with preference for hydrophobic substrates. Additionally, we solved crystal structures of this β-propeller enzyme separately with either phosphate, an inhibitor, or a mixture of GVL and products to define an active site where calcium-bound water and calcium-bound aspartic and glutamic acid residues make close contact with substrate and product. Our kinetic characterization of WT and mutant enzymes combined with structural insights inform a reaction mechanism that centers around activation of a calcium-bound water molecule promoted by general base catalysis and close contacts with substrate and a potential intermediate. Similarity of Rpa3624 with other β-propeller lactonases suggests this mechanism may be relevant for other members of this emerging class of versatile catalysts.
- Laboratory of Genetics, University of Wisconsin-Madison, Madison, Wisconsin, USA; Department of Energy Great Lakes Bioenergy Research Center, Madison, Wisconsin, USA; Wisconsin Energy Institute, University of Wisconsin-Madison, Madison, Wisconsin, USA.
Organizational Affiliation: