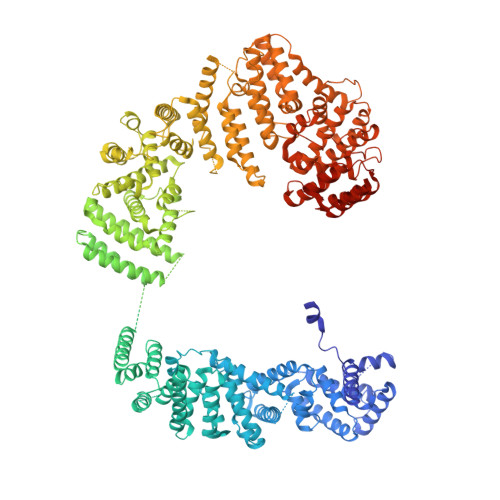
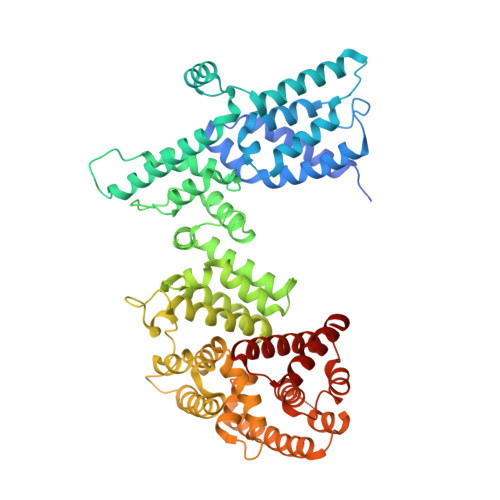
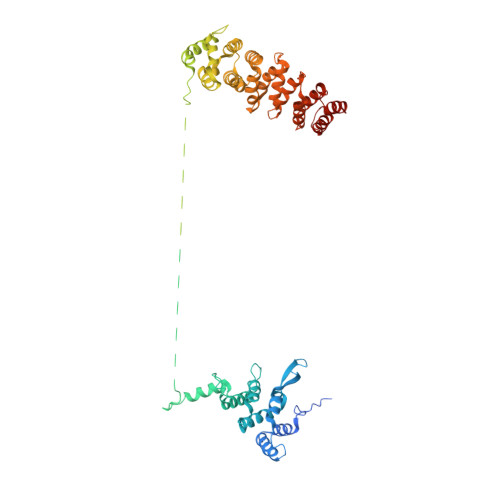
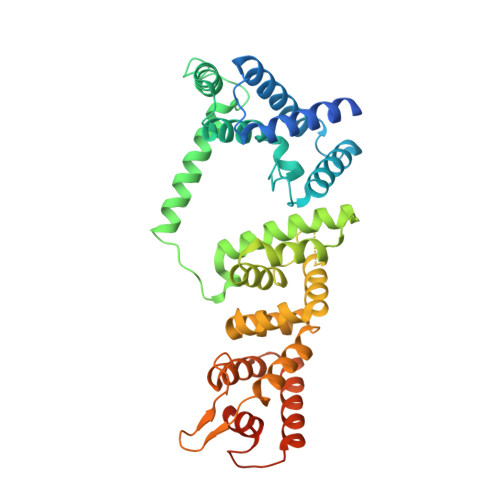
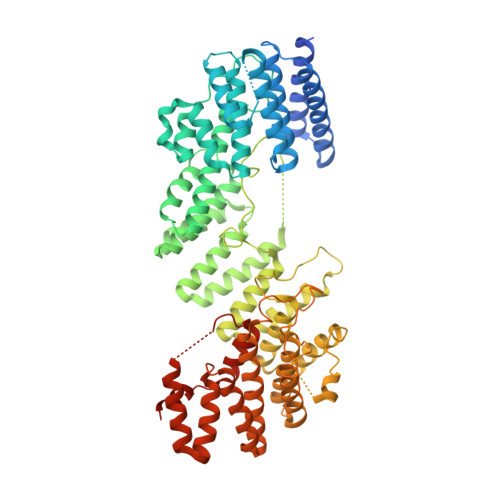
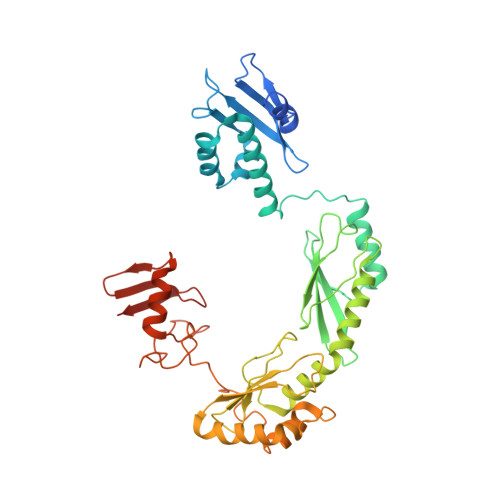
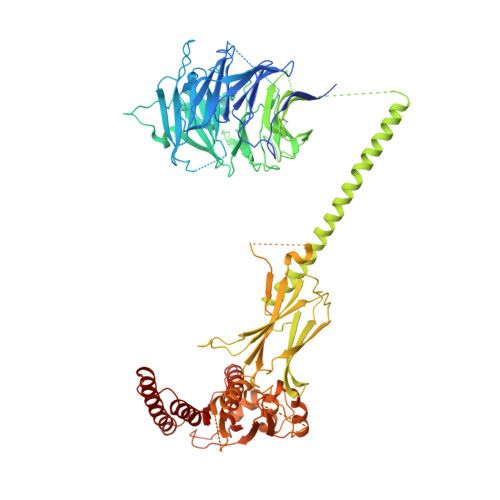
Structure of the FA core ubiquitin ligase closing the ID clamp on DNA.
Wang, S., Wang, R., Peralta, C., Yaseen, A., Pavletich, N.P.(2021) Nat Struct Mol Biol 28: 300-309
- PubMed: 33686268 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-021-00568-8
- Primary Citation Related Structures:
7KZP, 7KZQ, 7KZR, 7KZS, 7KZT, 7KZV - PubMed Abstract:
The Fanconi anemia (FA) pathway is essential for the repair of DNA interstrand crosslinks. Central to the pathway is the FA core complex, a ubiquitin ligase of nine subunits that monoubiquitinates the FANCI-FANCD2 (ID) DNA clamp. The 3.1 Å structure of the 1.1-MDa human FA core complex, described here, reveals an asymmetric assembly with two copies of all but the FANCC, FANCE and FANCF subunits. The asymmetry is crucial, as it prevents the binding of a second FANCC-FANCE-FANCF subcomplex that inhibits the recruitment of the UBE2T ubiquitin conjugating enzyme, and instead creates an ID binding site. A single active site then ubiquitinates FANCD2 and FANCI sequentially. We also present the 4.2-Å structures of the human core-UBE2T-ID-DNA complex in three conformations captured during monoubiquitination. They reveal the core-UBE2T complex remodeling the ID-DNA complex, closing the clamp on the DNA before ubiquitination. Monoubiquitination then prevents clamp opening after release from the core.
- Structural Biology Program, Memorial Sloan Kettering Cancer Center, New York, NY, USA.
Organizational Affiliation: