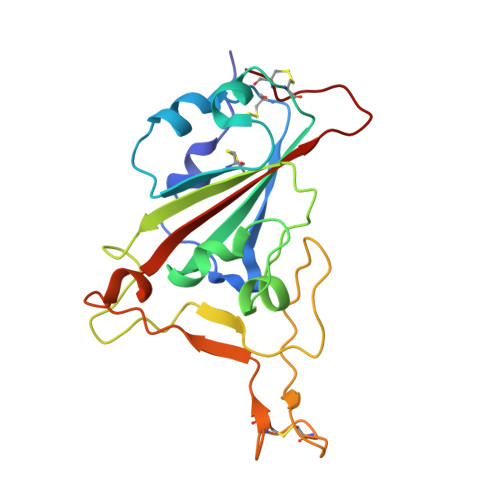
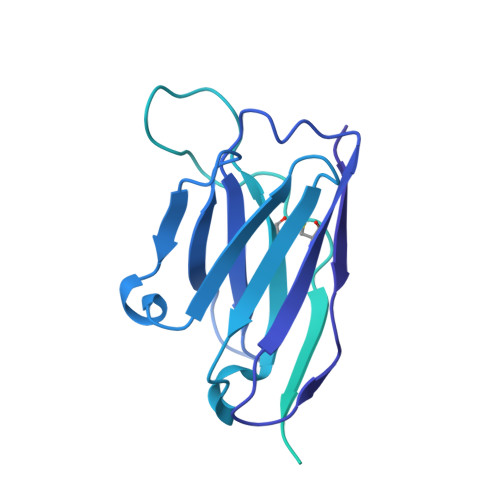
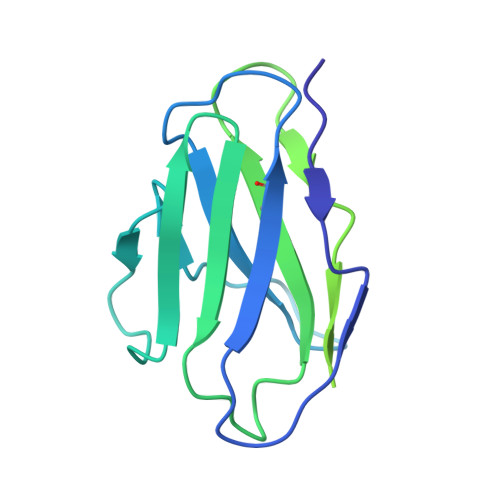
Three epitope-distinct human antibodies from RenMab mice neutralize SARS-CoV-2 and cooperatively minimize the escape of mutants.
Nie, J., Xie, J., Liu, S., Wu, J., Liu, C., Li, J., Liu, Y., Wang, M., Zhao, H., Zhang, Y., Yao, J., Chen, L., Shen, Y., Yang, Y., Wang, H.W., Wang, Y., Huang, W.(2021) Cell Discov 7: 53-53
- PubMed: 34285195 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41421-021-00292-z
- Primary Citation Related Structures:
7E39, 7E3B, 7E3C - PubMed Abstract:
Coronavirus disease 2019 (COVID-19), a pandemic disease caused by the newly emerging severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has caused more than 3.8 million deaths to date. Neutralizing antibodies are effective therapeutic measures. However, many naturally occurring mutations at the receptor-binding domain (RBD) have emerged, and some of them can evade existing neutralizing antibodies. Here, we utilized RenMab, a novel mouse carrying the entire human antibody variable region, for neutralizing antibody discovery. We obtained several potent RBD-blocking antibodies and categorized them into four distinct groups by epitope mapping. We determined the involved residues of the epitope of three representative antibodies by cryo-electron microscopy (Cryo-EM) studies. Moreover, we performed neutralizing experiments with 50 variant strains with single or combined mutations and found that the mixing of three epitope-distinct antibodies almost eliminated the mutant escape. Our study provides a sound basis for the rational design of fully human antibody cocktails against SARS-CoV-2 and pre-emergent coronaviral threats.
- Division of HIV/AIDS and Sex-transmitted Virus Vaccines, Institute for Biological Product Control, National Institutes for Food and Drug Control (NIFDC) and WHO Collaborating Center for Standardization and Evaluation of Biologicals, Beijing, China.
Organizational Affiliation: