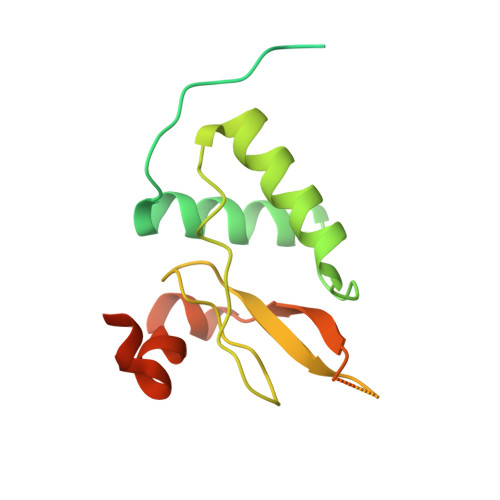
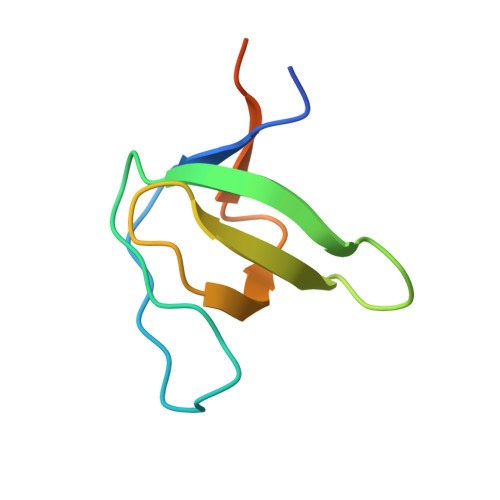
Synergy and allostery in ligand binding by HIV-1 Nef.
Aldehaiman, A., Momin, A.A., Restouin, A., Wang, L., Shi, X., Aljedani, S., Opi, S., Lugari, A., Shahul Hameed, U.F., Ponchon, L., Morelli, X., Huang, M., Dumas, C., Collette, Y., Arold, S.T.(2021) Biochem J 478: 1525-1545
- PubMed: 33787846 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1042/BCJ20201002
- Primary Citation Related Structures:
3H0F, 3H0H, 3H0I, 4D8D, 6IPY, 6IPZ, 7D7S - PubMed Abstract:
The Nef protein of human and simian immunodeficiency viruses boosts viral pathogenicity through its interactions with host cell proteins. By combining the polyvalency of its large unstructured regions with the binding selectivity and strength of its folded core domain, Nef can associate with many different host cell proteins, thereby disrupting their functions. For example, the combination of a linear proline-rich motif and hydrophobic core domain surface allows Nef to bind tightly and specifically to SH3 domains of Src family kinases. We investigated whether the interplay between Nef's flexible regions and its core domain could allosterically influence ligand selection. We found that the flexible regions can associate with the core domain in different ways, producing distinct conformational states that alter the way in which Nef selects for SH3 domains and exposes some of its binding motifs. The ensuing crosstalk between ligands might promote functionally coherent Nef-bound protein ensembles by synergizing certain subsets of ligands while excluding others. We also combined proteomic and bioinformatics analyses to identify human proteins that select SH3 domains in the same way as Nef. We found that only 3% of clones from a whole-human fetal library displayed Nef-like SH3 selectivity. However, in most cases, this selectivity appears to be achieved by a canonical linear interaction rather than by a Nef-like 'tertiary' interaction. Our analysis supports the contention that Nef's mode of hijacking SH3 domains is a virus-specific adaptation with no or very few cellular counterparts. Thus, the Nef tertiary binding surface is a promising virus-specific drug target.
- King Abdullah University of Science and Technology (KAUST), Computational Bioscience Research Center (CBRC), Biological and Environmental Science and Engineering (BESE), Thuwal 23955-6900, Saudi Arabia.
Organizational Affiliation: