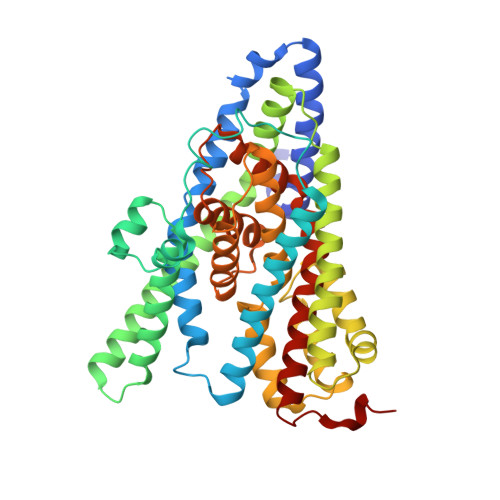
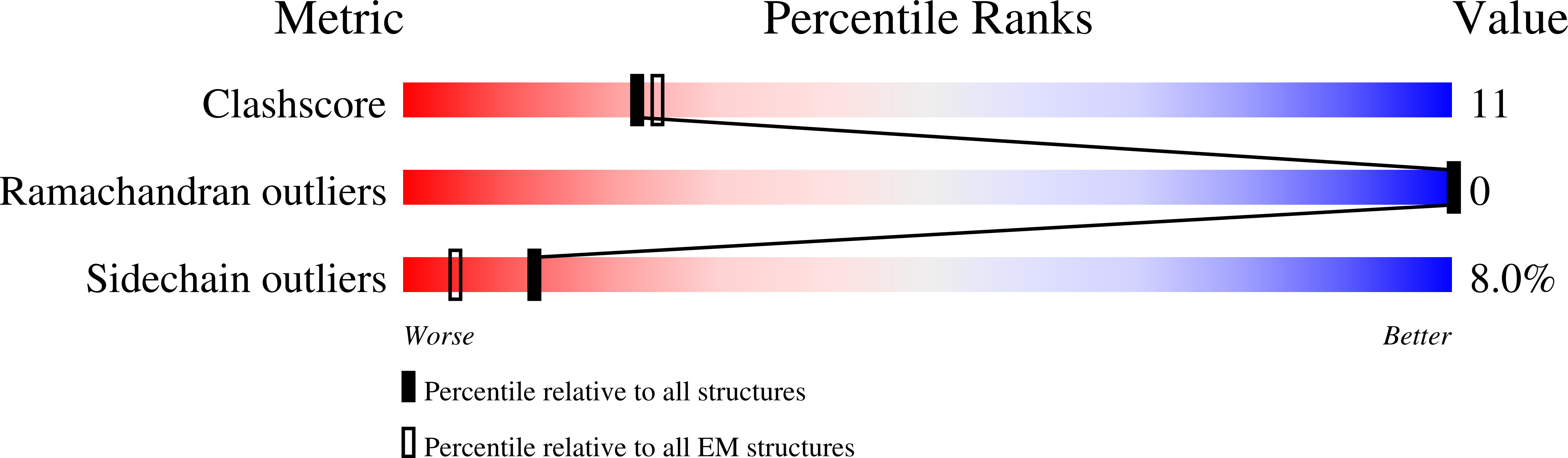Structural ensemble of a glutamate transporter homologue in lipid nanodisc environment.
Arkhipova, V., Guskov, A., Slotboom, D.J.(2020) Nat Commun 11: 998-998
- PubMed: 32081874 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-020-14834-8
- Primary Citation Related Structures:
6XWN, 6XWO, 6XWP, 6XWQ, 6XWR - PubMed Abstract:
Glutamate transporters are cation-coupled secondary active membrane transporters that clear the neurotransmitter L-glutamate from the synaptic cleft. These transporters are homotrimers, with each protomer functioning independently by an elevator-type mechanism, in which a mobile transport domain alternates between inward- and outward-oriented states. Using single-particle cryo-EM we have determined five structures of the glutamate transporter homologue Glt Tk , a Na + - L-aspartate symporter, embedded in lipid nanodiscs. Dependent on the substrate concentrations used, the protomers of the trimer adopt a variety of asymmetrical conformations, consistent with the independent movement. Six of the 15 resolved protomers are in a hitherto elusive state of the transport cycle in which the inward-facing transporters are loaded with Na + ions. These structures explain how substrate-leakage is prevented - a strict requirement for coupled transport. The belt protein of the lipid nanodiscs bends around the inward oriented protomers, suggesting that membrane deformations occur during transport.
- Groningen Biomolecular Sciences and Biotechnology Institute (GBB), University of Groningen, Nijenborgh 4, 9747AG, Groningen, The Netherlands.
Organizational Affiliation: