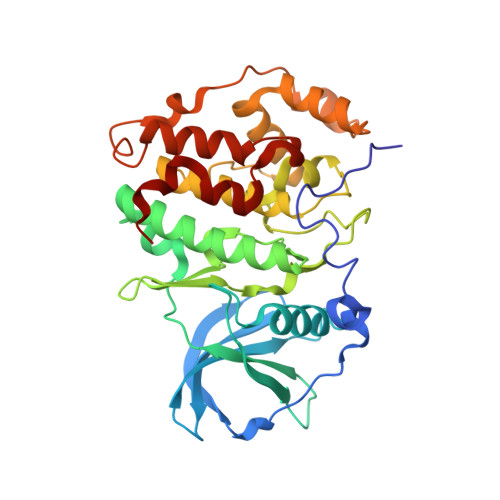
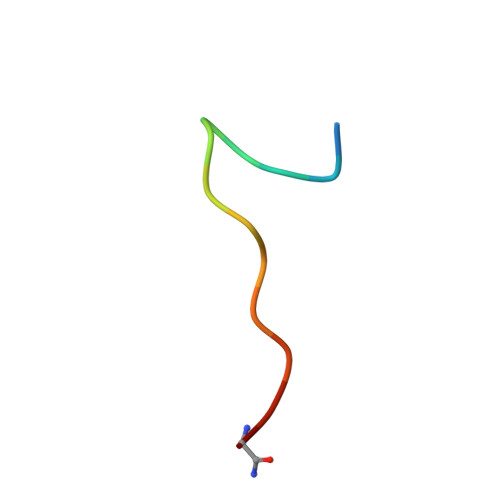
Efficient development of stable and highly functionalised peptides targeting the CK2 alpha /CK2 beta protein-protein interaction.
Iegre, J., Brear, P., Baker, D.J., Tan, Y.S., Atkinson, E.L., Sore, H.F., O' Donovan, D.H., Verma, C.S., Hyvonen, M., Spring, D.R.(2019) Chem Sci 10: 5056-5063
- PubMed: 31183056 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1039/c9sc00798a
- Primary Citation Related Structures:
6Q38, 6Q4Q - PubMed Abstract:
The discovery of new Protein-Protein Interaction (PPI) modulators is currently limited by the difficulties associated with the design and synthesis of selective small molecule inhibitors. Peptides are a potential solution for disrupting PPIs; however, they typically suffer from poor stability in vivo and limited tissue penetration hampering their wide spread use as new chemical biology tools and potential therapeutics. In this work, a combination of CuAAC chemistry, molecular modelling, X-ray crystallography, and biological validation allowed us to develop highly functionalised peptide PPI inhibitors of the protein CK2. The lead peptide, CAM7117 , prevents the formation of the holoenzyme assembly in vitro , slows down proliferation, induces apoptosis in cancer cells and is stable in human serum. CAM7117 could aid the development of novel CK2 inhibitors acting at the interface and help to fully understand the intracellular pathways involving CK2. Importantly, the approach adopted herein could be applied to many PPI targets and has the potential to ease the study of PPIs by efficiently providing access to functionalised peptides.
- Department of Chemistry , University of Cambridge , Lensfield Road , CB2 1EW , Cambridge , UK . Email: spring@ch.cam.ac.uk.
Organizational Affiliation: