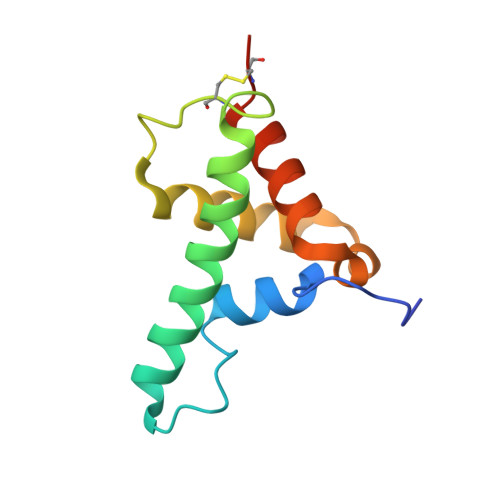
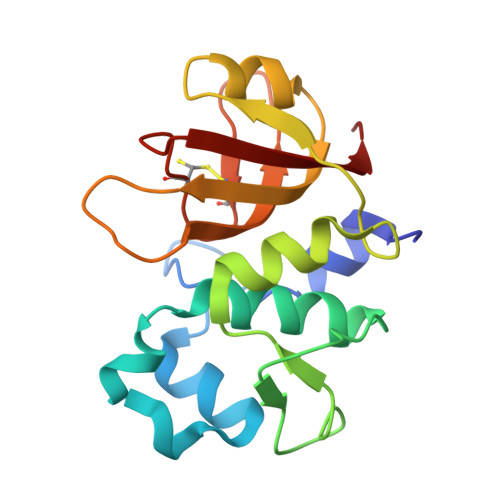
Crystal structure of the Agrobacterium tumefaciens type VI effector-immunity complex.
Fukuhara, S., Nakane, T., Yamashita, K., Ishii, R., Ishitani, R., Nureki, O.(2018) Acta Crystallogr F Struct Biol Commun 74: 810-816
- PubMed: 30511676 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S2053230X18016369
- Primary Citation Related Structures:
6IJE, 6IJF - PubMed Abstract:
The type VI secretion system (T6SS) comprises needle-shaped multisubunit complexes that play a role in the microbial defense systems of Gram-negative bacteria. Some Gram-negative bacteria harboring a T6SS deliver toxic effector proteins into the cytoplasm or periplasm of competing bacteria in order to lyse and kill them. To avoid self-cell disruption, these bacteria have cognate immunity proteins that inhibit their toxic effector proteins. T6SS amidase effector protein 4 (Tae4) and T6SS amidase immunity protein 4 (Tai4) are a representative of the toxic effector-immunity pairs of the T6SS. Here, the three-dimensional structures of Tai4 and the Tae4-Tai4 complex from Agrobacterium tumefaciens are reported at 1.55 and 1.9 Å resolution, respectively. A structural comparison with other Tae4-Tai4 homologs revealed similarities and differences in the catalytic and inhibitory mechanisms among the Tae4 and Tai4 family proteins.
- Department of Biological Sciences, Graduate School of Science, The University of Tokyo, 7-3-1 Hongo, Bunkyo-ku, Tokyo 113-0033, Japan.
Organizational Affiliation: