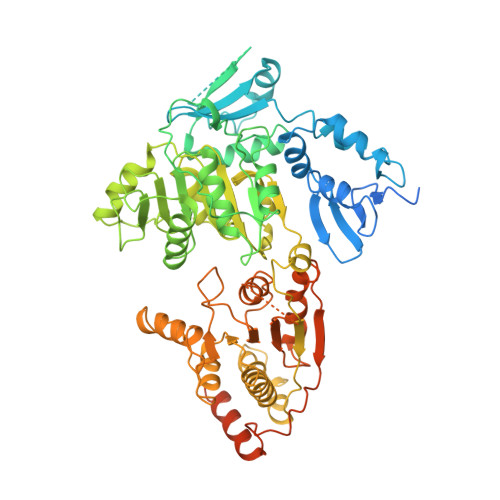
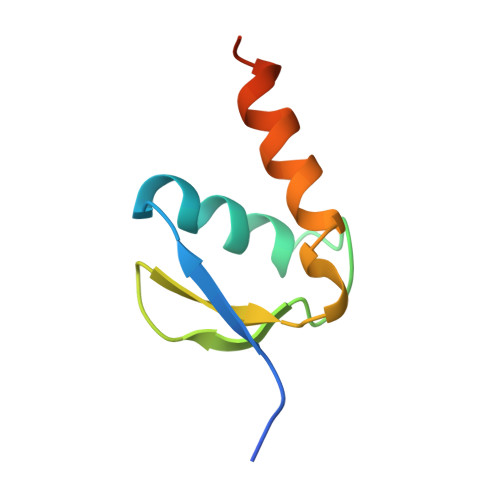
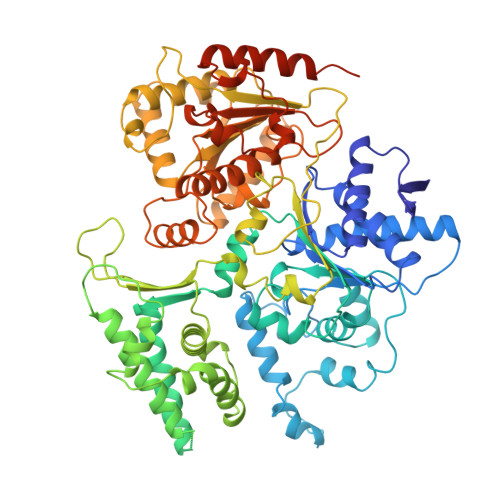
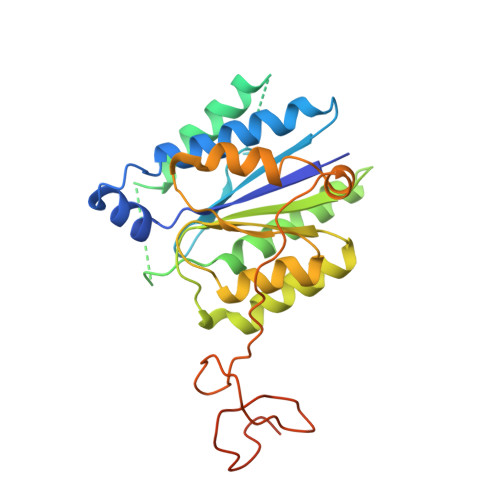
Structural basis of TFIIH activation for nucleotide excision repair.
Kokic, G., Chernev, A., Tegunov, D., Dienemann, C., Urlaub, H., Cramer, P.(2019) Nat Commun 10: 2885-2885
- PubMed: 31253769 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-019-10745-5
- Primary Citation Related Structures:
6RO4 - PubMed Abstract:
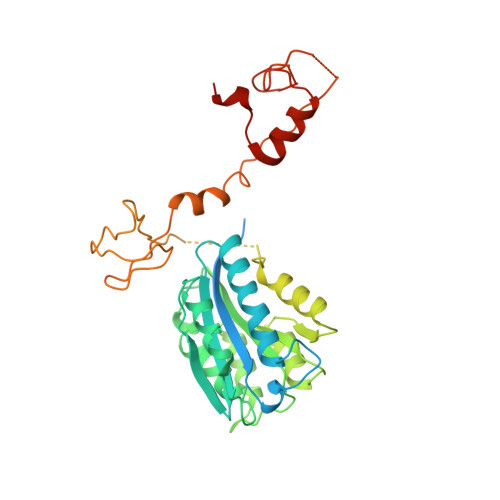
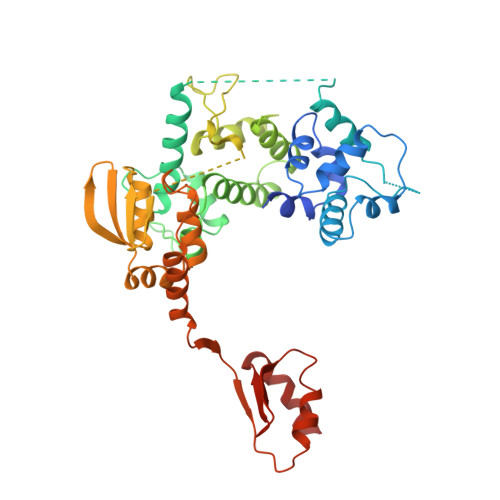
Nucleotide excision repair (NER) is the major DNA repair pathway that removes UV-induced and bulky DNA lesions. There is currently no structure of NER intermediates, which form around the large multisubunit transcription factor IIH (TFIIH). Here we report the cryo-EM structure of an NER intermediate containing TFIIH and the NER factor XPA. Compared to its transcription conformation, the TFIIH structure is rearranged such that its ATPase subunits XPB and XPD bind double- and single-stranded DNA, consistent with their translocase and helicase activities, respectively. XPA releases the inhibitory kinase module of TFIIH, displaces a 'plug' element from the DNA-binding pore in XPD, and together with the NER factor XPG stimulates XPD activity. Our results explain how TFIIH is switched from a transcription to a repair factor, and provide the basis for a mechanistic analysis of the NER pathway.
- Department of Molecular Biology, Max Planck Institute for Biophysical Chemistry, Am Fassberg 11, 37077, Göttingen, Germany.
Organizational Affiliation: