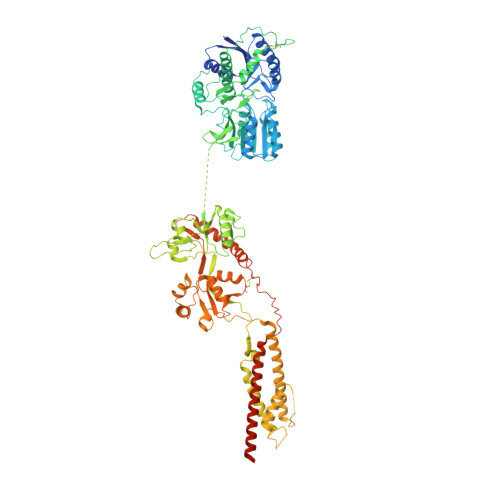
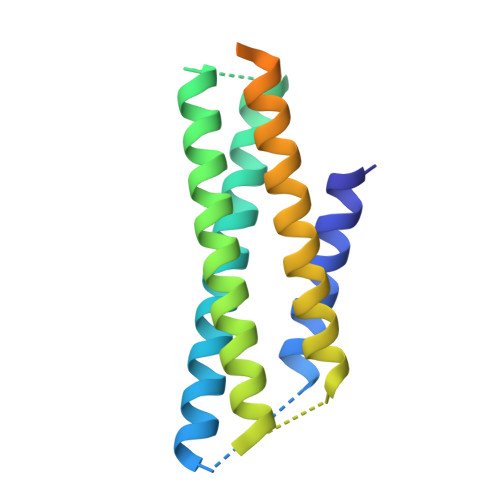
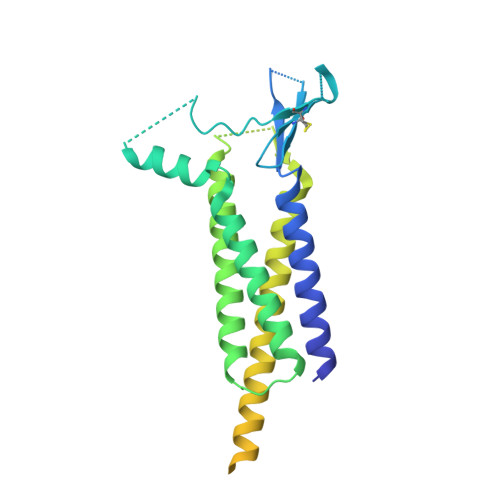
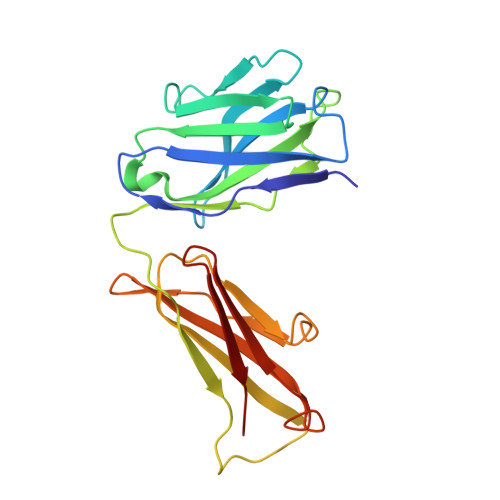
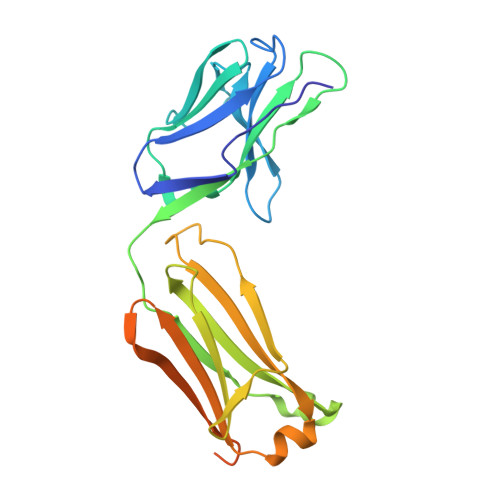
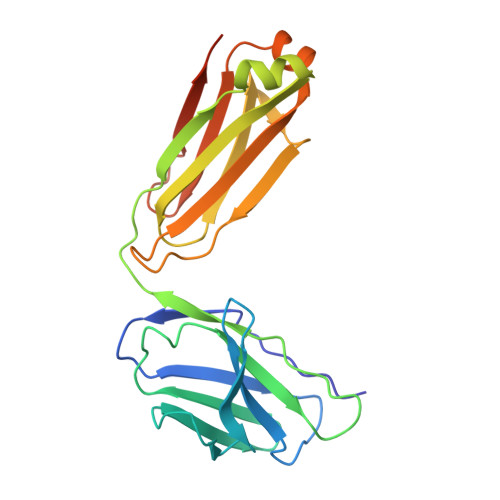
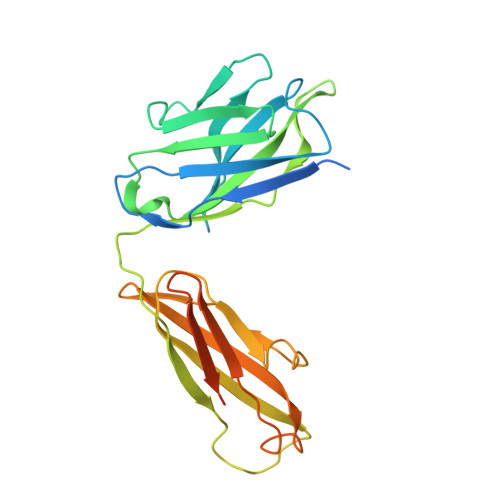
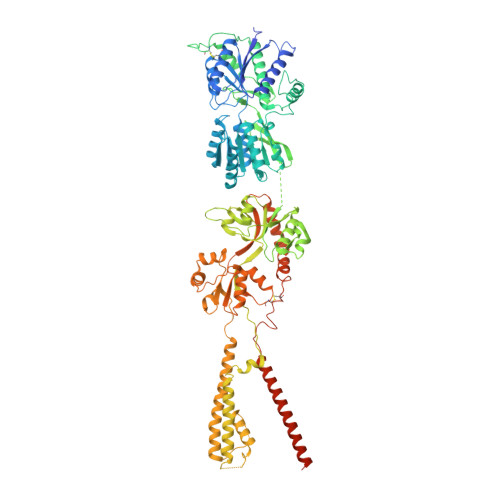Architecture and subunit arrangement of native AMPA receptors elucidated by cryo-EM.
Zhao, Y., Chen, S., Swensen, A.C., Qian, W.J., Gouaux, E.(2019) Science 364: 355-362
- PubMed: 30975770 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/science.aaw8250
- Primary Citation Related Structures:
6NJL, 6NJM, 6NJN - PubMed Abstract:
Glutamate-gated AMPA receptors mediate the fast component of excitatory signal transduction at chemical synapses throughout all regions of the mammalian brain. AMPA receptors are tetrameric assemblies composed of four subunits, GluA1-GluA4. Despite decades of study, the subunit composition, subunit arrangement, and molecular structure of native AMPA receptors remain unknown. Here we elucidate the structures of 10 distinct native AMPA receptor complexes by single-particle cryo-electron microscopy (cryo-EM). We find that receptor subunits are arranged nonstochastically, with the GluA2 subunit preferentially occupying the B and D positions of the tetramer and with triheteromeric assemblies comprising a major population of native AMPA receptors. Cryo-EM maps define the structure for S2-M4 linkers between the ligand-binding and transmembrane domains, suggesting how neurotransmitter binding is coupled to ion channel gating.
- Vollum Institute, Oregon Health and Science University, 3181 SW Sam Jackson Park Road, Portland, OR 97239, USA.
Organizational Affiliation: