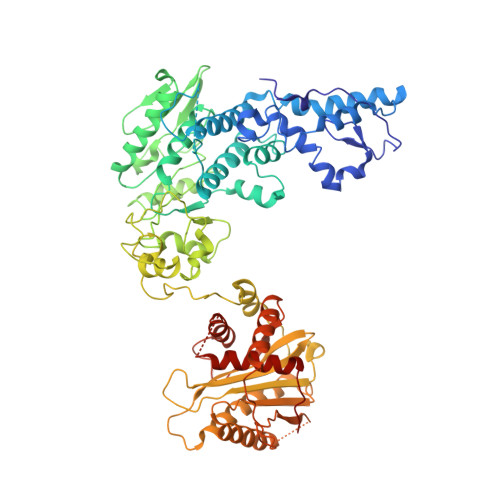
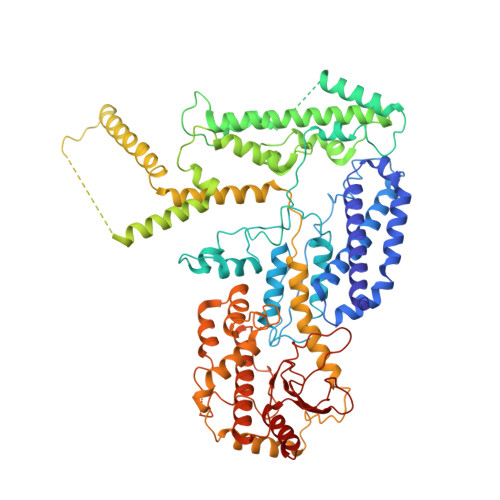
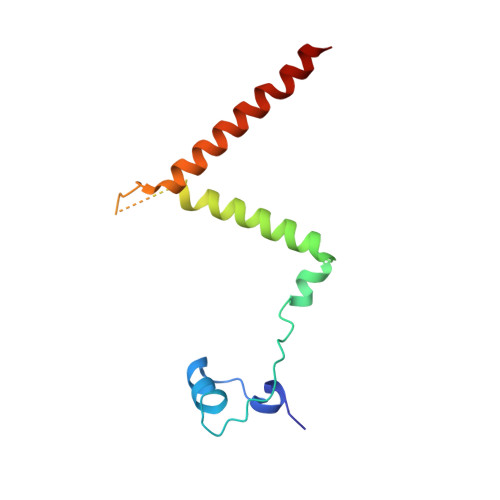
Architecture of the herpesvirus genome-packaging complex and implications for DNA translocation.
Yang, Y., Yang, P., Wang, N., Chen, Z., Su, D., Zhou, Z.H., Rao, Z., Wang, X.(2020) Protein Cell 11: 339-351
- PubMed: 32328903 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1007/s13238-020-00710-0
- Primary Citation Related Structures:
6M5R, 6M5S, 6M5T, 6M5U, 6M5V - PubMed Abstract:
Genome packaging is a fundamental process in a viral life cycle and a prime target of antiviral drugs. Herpesviruses use an ATP-driven packaging motor/terminase complex to translocate and cleave concatemeric dsDNA into procapsids but its molecular architecture and mechanism are unknown. We report atomic structures of a herpesvirus hexameric terminase complex in both the apo and ADP•BeF3-bound states. Each subunit of the hexameric ring comprises three components-the ATPase/terminase pUL15 and two regulator/fixer proteins, pUL28 and pUL33-unlike bacteriophage terminases. Distal to the nuclease domains, six ATPase domains form a central channel with conserved basic-patches conducive to DNA binding and trans-acting arginine fingers are essential to ATP hydrolysis and sequential DNA translocation. Rearrangement of the nuclease domains mediated by regulatory domains converts DNA translocation mode to cleavage mode. Our structures favor a sequential revolution model for DNA translocation and suggest mechanisms for concerted domain rearrangements leading to DNA cleavage.
- CAS Key Laboratory of Infection and Immunity, National Laboratory of Macromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, 100101, China.
Organizational Affiliation: