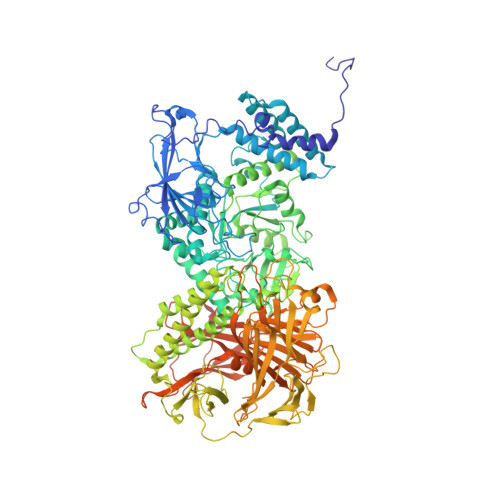
Cryo-EM structure of fission yeast tetrameric alpha-mannosidase Ams1.
Zhang, J., Wang, Y.Y., Du, L.L., Ye, K.(2020) FEBS Open Bio 10: 2437-2451
- PubMed: 32981237
- DOI: https://doi.org/10.1002/2211-5463.12988
- Primary Citation of Related Structures:
6LZ1 - PubMed Abstract:
Fungal α-mannosidase Ams1 and its mammalian homolog MAN2C1 hydrolyze terminal α-linked mannoses in free oligosaccharides released from misfolded glycoproteins or lipid-linked oligosaccharide donors. Ams1 is transported by selective autophagy into vacuoles. Here, we determine the tetrameric structure of Ams1 from the fission yeast Schizosaccharomyces pombe at 3.2 Å resolution by cryo-electron microscopy. Distinct from a low resolution structure of S. cerevisiae Ams1, S. pombe Ams1 has a prominent N-terminal tail that mediates tetramerization and an extra β-sheet domain. Ams1 shares a conserved active site with other enzymes in glycoside hydrolase family 38, to which Ams1 belongs, but contains extra N-terminal domains involved in tetramerization. The atomic structure of Ams1 reported here will aid understanding of its enzymatic activity and transport mechanism.
- Key Laboratory of RNA Biology, CAS Center for Excellence in Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.
Organizational Affiliation: