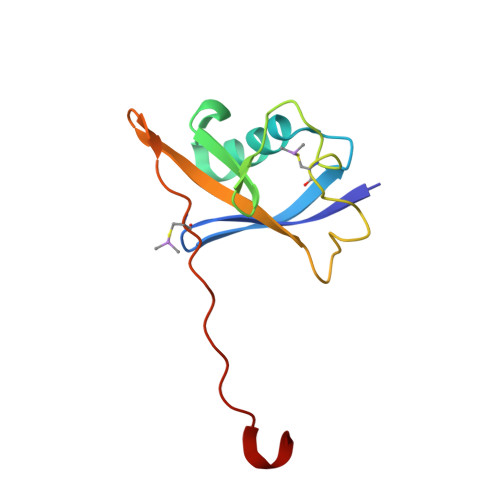
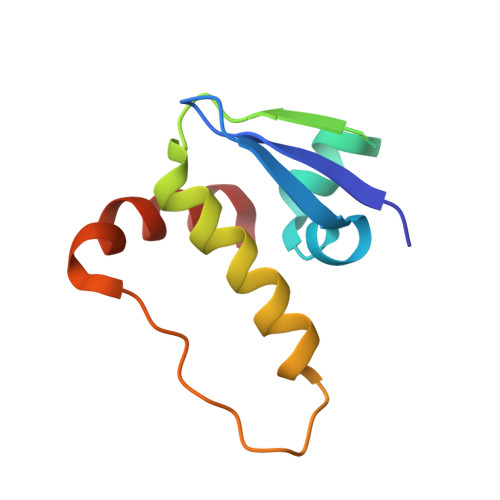
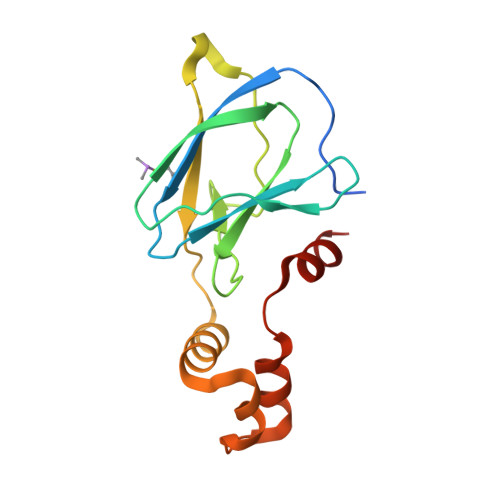
Thioamide substitution to probe the hydroxyproline recognition of VHL ligands.
Soares, P., Lucas, X., Ciulli, A.(2018) Bioorg Med Chem 26: 2992-2995
- PubMed: 29650462 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.bmc.2018.03.034
- Primary Citation Related Structures:
6FMI, 6FMJ, 6FMK - PubMed Abstract:
Thioamide substitution influences hydrogen bond and n → π ∗ interactions involved in the conformational stability of protein secondary structures and oligopeptides. Hydroxyproline is the key recognition element of small molecules targeting the von Hippel-Lindau (VHL) E3 ligase, which are of interest as probes of hypoxia signaling and ligands for PROTAC conjugation. We hypothesized that VHL ligands could be a privileged model system to evaluate the contribution of these interactions to protein:ligand complex formation. Herein we report the synthesis of VHL ligands bearing thioamide substitutions at the central hydroxyproline moiety, and characterize their binding by fluorescence polarization, isothermal titration calorimetry, X-ray crystallography and molecular modeling. In spite of a conserved binding mode, the substitution pattern had a pronounced impact on the ligand affinities. Together the results underscore the role of hydrogen bond and n → π ∗ interactions in fine tuning hydroxyproline recognition by VHL.
- Division of Biological Chemistry and Drug Discovery, School of Life Sciences, University of Dundee, Dow Street, Dundee DD1 5EH, Scotland, UK.
Organizational Affiliation: