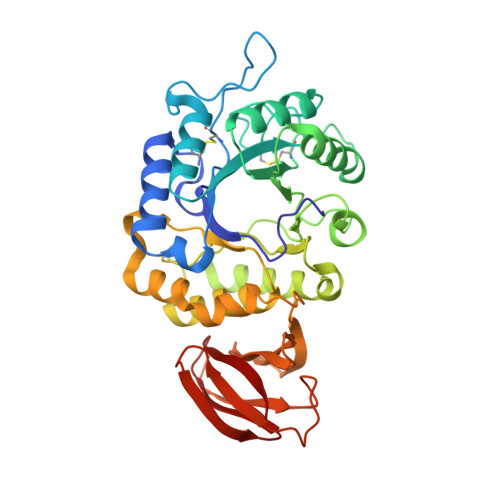
Nicotiana benthamianaalpha-galactosidase A1.1 can functionally complement human alpha-galactosidase A deficiency associated with Fabry disease.
Kytidou, K., Beekwilder, J., Artola, M., van Meel, E., Wilbers, R.H.P., Moolenaar, G.F., Goosen, N., Ferraz, M.J., Katzy, R., Voskamp, P., Florea, B.I., Hokke, C.H., Overkleeft, H.S., Schots, A., Bosch, D., Pannu, N., Aerts, J.M.F.G.(2018) J Biological Chem 293: 10042-10058
- PubMed: 29674318 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.RA118.001774
- Primary Citation Related Structures:
6F4C - PubMed Abstract:
α-Galactosidases (EC 3.2.1.22) are retaining glycosidases that cleave terminal α-linked galactose residues from glycoconjugate substrates. α-Galactosidases take part in the turnover of cell wall-associated galactomannans in plants and in the lysosomal degradation of glycosphingolipids in animals. Deficiency of human α-galactosidase A (α-Gal A) causes Fabry disease (FD), a heritable, X-linked lysosomal storage disorder, characterized by accumulation of globotriaosylceramide (Gb3) and globotriaosylsphingosine (lyso-Gb3). Current management of FD involves enzyme-replacement therapy (ERT). An activity-based probe (ABP) covalently labeling the catalytic nucleophile of α-Gal A has been previously designed to study α-galactosidases for use in FD therapy. Here, we report that this ABP labels proteins in Nicotiana benthamiana leaf extracts, enabling the identification and biochemical characterization of an N. benthamiana α-galactosidase we name here A1.1 (gene accession ID GJZM-1660). The transiently overexpressed and purified enzyme was a monomer lacking N -glycans and was active toward 4-methylumbelliferyl-α-d-galactopyranoside substrate ( K m = 0.17 mm) over a broad pH range. A1.1 structural analysis by X-ray crystallography revealed marked similarities with human α-Gal A, even including A1.1's ability to hydrolyze Gb3 and lyso-Gb3, which are not endogenous in plants. Of note, A1.1 uptake into FD fibroblasts reduced the elevated lyso-Gb3 levels in these cells, consistent with A1.1 delivery to lysosomes as revealed by confocal microscopy. The ease of production and the features of A1.1, such as stability over a broad pH range, combined with its capacity to degrade glycosphingolipid substrates, warrant further examination of its value as a potential therapeutic agent for ERT-based FD management.
- From the Departments of Medical Biochemistry.
Organizational Affiliation: