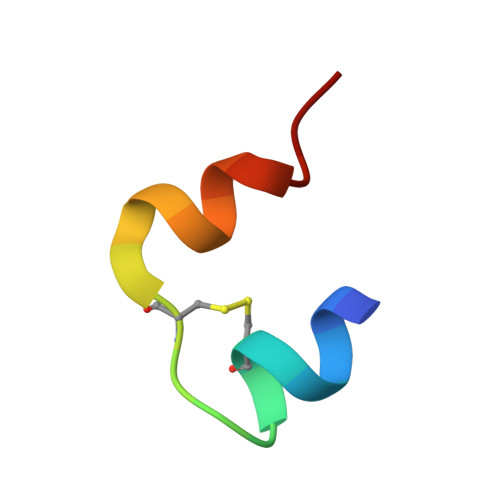
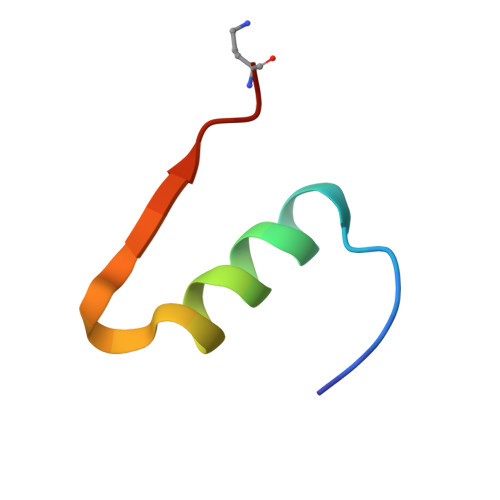
Structure-based stabilization of insulin as a therapeutic protein assembly via enhanced aromatic-aromatic interactions.
Rege, N.K., Wickramasinghe, N.P., Tustan, A.N., Phillips, N.F.B., Yee, V.C., Ismail-Beigi, F., Weiss, M.A.(2018) J Biological Chem 293: 10895-10910
- PubMed: 29880646 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.RA118.003650
- Primary Citation Related Structures:
6CK2 - PubMed Abstract:
Key contributions to protein structure and stability are provided by weakly polar interactions, which arise from asymmetric electronic distributions within amino acids and peptide bonds. Of particular interest are aromatic side chains whose directional π-systems commonly stabilize protein interiors and interfaces. Here, we consider aromatic-aromatic interactions within a model protein assembly: the dimer interface of insulin. Semi-classical simulations of aromatic-aromatic interactions at this interface suggested that substitution of residue Tyr B26 by Trp would preserve native structure while enhancing dimerization (and hence hexamer stability). The crystal structure of a [Trp B26 ]insulin analog (determined as a T 3 R f 3 zinc hexamer at a resolution of 2.25 Å) was observed to be essentially identical to that of WT insulin. Remarkably and yet in general accordance with theoretical expectations, spectroscopic studies demonstrated a 150-fold increase in the in vitro lifetime of the variant hexamer, a critical pharmacokinetic parameter influencing design of long-acting formulations. Functional studies in diabetic rats indeed revealed prolonged action following subcutaneous injection. The potency of the Trp B26 -modified analog was equal to or greater than an unmodified control. Thus, exploiting a general quantum-chemical feature of protein structure and stability, our results exemplify a mechanism-based approach to the optimization of a therapeutic protein assembly.
- From the Departments of Biochemistry and.
Organizational Affiliation: