Catalytic mechanism of tyrosinase implied from the quinone formation on the Tyr98 residue of the caddie protein
Matoba, Y., Sugiyama, M.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Tyrosinase | 281 | Streptomyces castaneoglobisporus | Mutation(s): 0 Gene Names: tyrC EC: 1.14.18.1 | 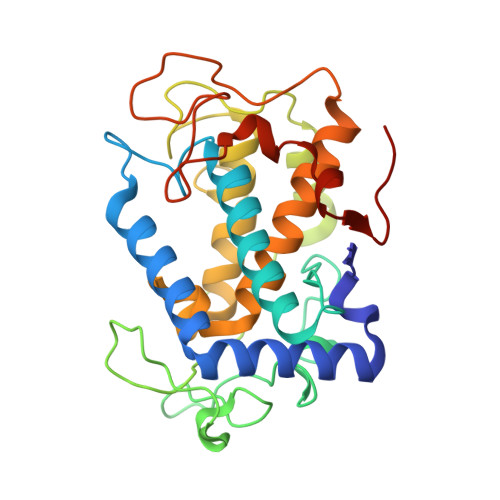 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q83WS2 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| MelC | 134 | Streptomyces castaneoglobisporus | Mutation(s): 0 Gene Names: orf378 | 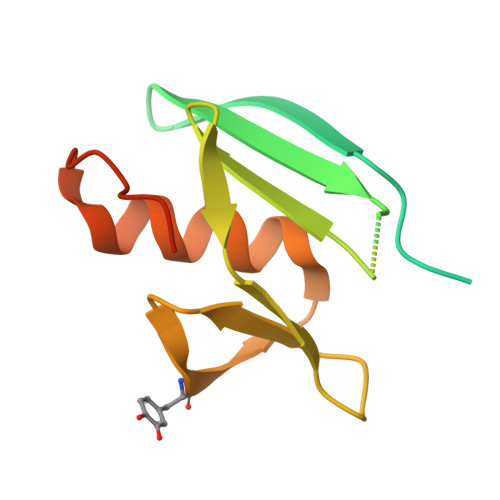 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q83WS1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CU Download:Ideal Coordinates CCD File | C [auth A], D [auth A], E [auth A], J [auth B] | COPPER (II) ION Cu JPVYNHNXODAKFH-UHFFFAOYSA-N |  | ||
| NO3 Download:Ideal Coordinates CCD File | G [auth A], H [auth A], I [auth A], K [auth B], L [auth B] | NITRATE ION N O3 NHNBFGGVMKEFGY-UHFFFAOYSA-N |  | ||
| PER Download:Ideal Coordinates CCD File | F [auth A] | PEROXIDE ION O2 ANAIPYUSIMHBEL-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| DAH Query on DAH | B | L-PEPTIDE LINKING | C9 H11 N O4 |  | TYR |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 65.2 | α = 90 |
| b = 97.79 | β = 90 |
| c = 55.1 | γ = 90 |
| Software Name | Purpose |
|---|---|
| CNS | phasing |
| SHELXL-97 | refinement |
| MOSFLM | data reduction |
| SCALA | data scaling |
| Funding Organization | Location | Grant Number |
|---|---|---|
| KAKENHI | Japan | 25109530, 15H009470 |