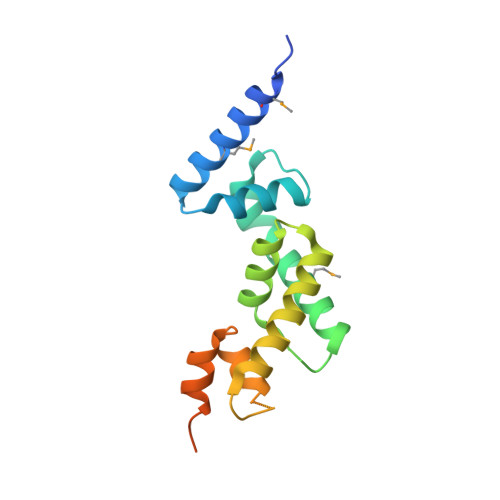
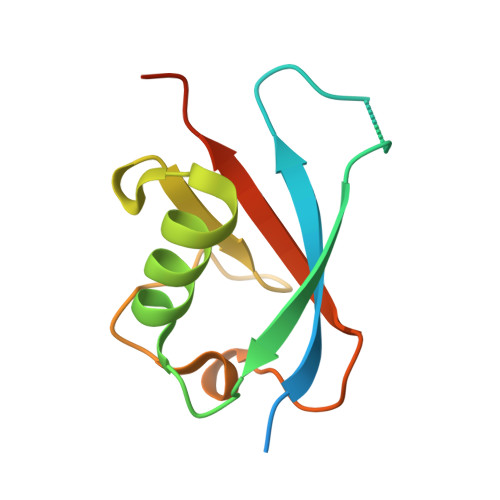
Structural Insights into SHARPIN-Mediated Activation of HOIP for the Linear Ubiquitin Chain Assembly
Liu, J., Wang, Y., Gong, Y., Fu, T., Hu, S., Zhou, Z., Pan, L.(2017) Cell Rep 21: 27-36
- PubMed: 28978479 Search on PubMed
- DOI: https://doi.org/10.1016/j.celrep.2017.09.031
- Primary Citation Related Structures:
5X0W - PubMed Abstract:
The linear ubiquitin chain assembly complex (LUBAC) is the sole identified E3 ligase complex that catalyzes the formation of linear ubiquitin chain, and it is composed of HOIP, HOIL-1L, and SHARPIN. The E3 activity of HOIP can be effectively activated by HOIL-1L or SHARPIN, deficiency of which leads to severe immune system disorders. However, the underlying mechanism governing the HOIP-SHARPIN interaction and the SHARPIN-mediated activation of HOIP remains elusive. Here, we biochemically and structurally demonstrate that the UBL domain of SHARPIN specifically binds to the UBA domain of HOIP and thereby associates with and activates HOIP. We further uncover that SHARPIN and HOIL-1L can separately or synergistically bind to distinct sites of HOIP UBA with induced allosteric effects and thereby facilitate the E2 loading of HOIP for its activation. Thus, our findings provide mechanistic insights into the assembly and activation of LUBAC.
- State Key Laboratory of Bioorganic and Natural Products Chemistry, Shanghai Institute of Organic Chemistry, University of Chinese Academy of Sciences, Chinese Academy of Sciences, 345 Lingling Road, Shanghai 200032, People's Republic of China.
Organizational Affiliation: