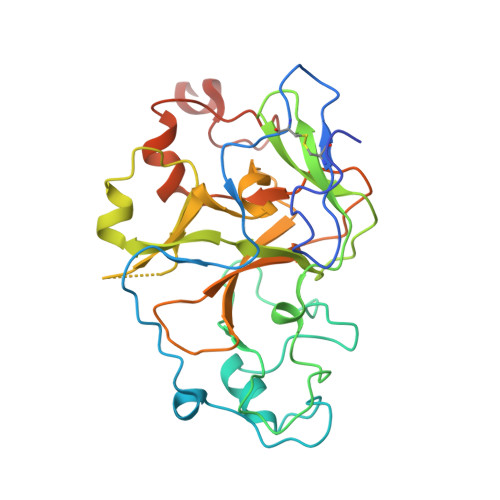
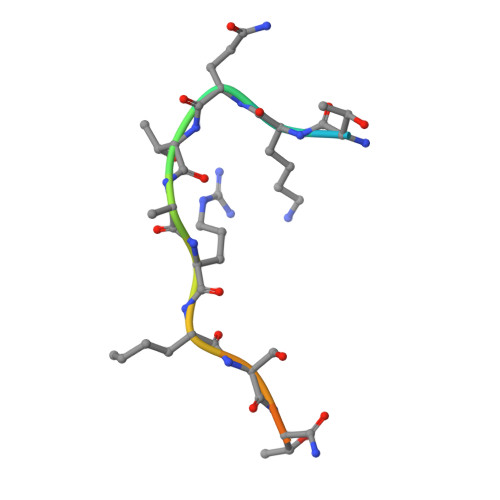
A histone H3K9M mutation traps histone methyltransferase Clr4 to prevent heterochromatin spreading.
Shan, C.M., Wang, J., Xu, K., Chen, H., Yue, J.X., Andrews, S., Moresco, J.J., Yates, J.R., Nagy, P.L., Tong, L., Jia, S.(2016) Elife 5
- PubMed: 27648579 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.17903
- Primary Citation Related Structures:
5T0K, 5T0M - PubMed Abstract:
Histone lysine-to-methionine (K-to-M) mutations are associated with multiple cancers, and they function in a dominant fashion to block the methylation of corresponding lysines on wild type histones. However, their mechanisms of function are controversial. Here we show that in fission yeast, introducing the K9M mutation into one of the three histone H3 genes dominantly blocks H3K9 methylation on wild type H3 across the genome. In addition, H3K9M enhances the interaction of histone H3 tail with the H3K9 methyltransferase Clr4 in a SAM (S-adenosyl-methionine)-dependent manner, and Clr4 is trapped at nucleation sites to prevent its spreading and the formation of large heterochromatin domains. We further determined the crystal structure of an H3K9M peptide in complex with human H3K9 methyltransferase G9a and SAM, which reveales that the methionine side chain had enhanced van der Waals interactions with G9a. Therefore, our results provide a detailed mechanism by which H3K9M regulates H3K9 methylation.
- Department of Biological Sciences, Columbia University, New York, United States.
Organizational Affiliation: