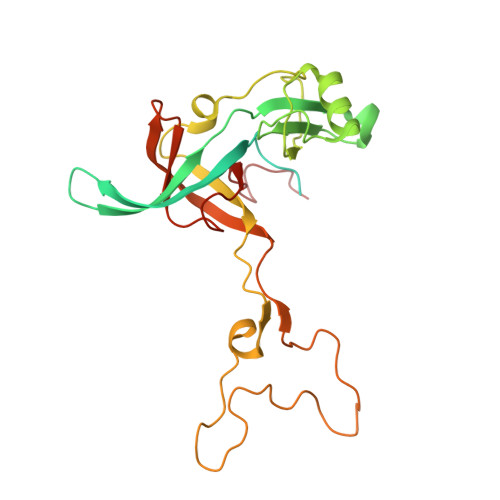
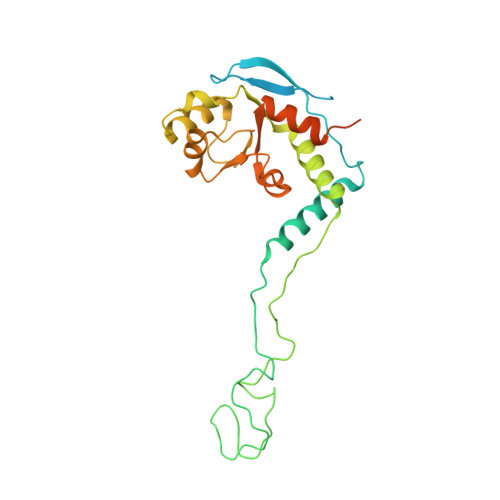
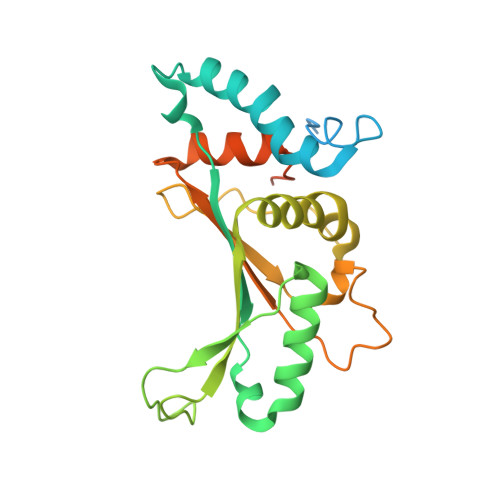
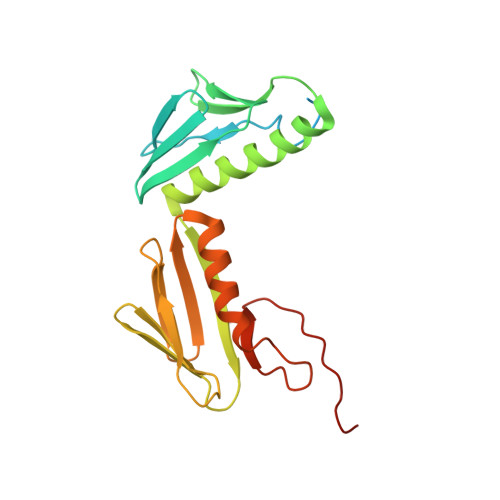
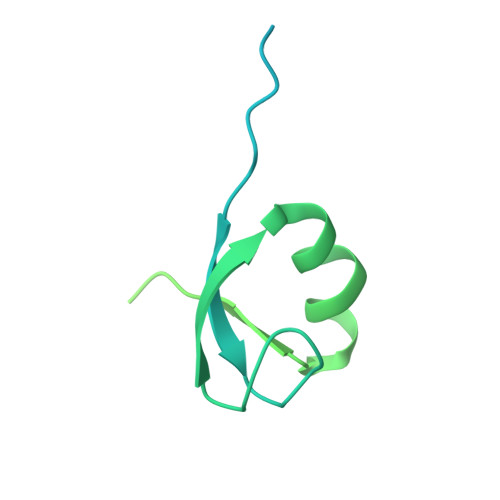
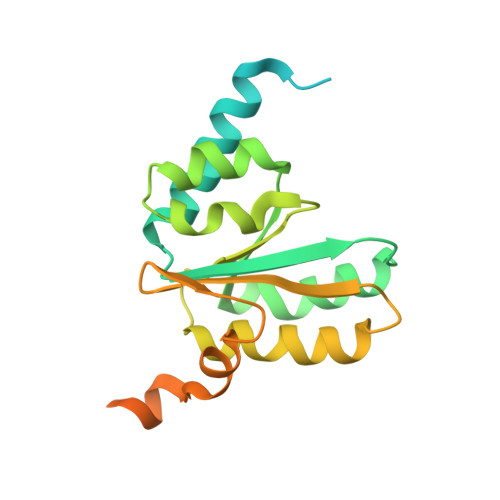
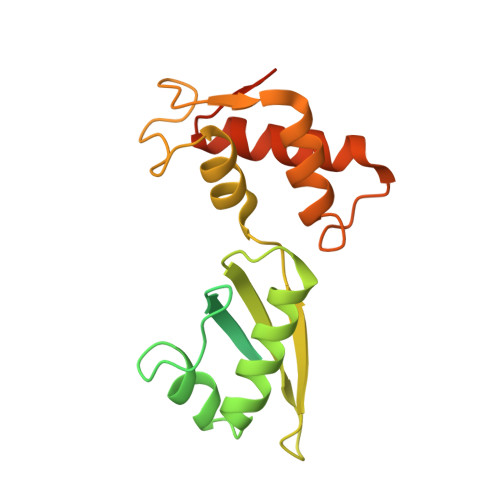
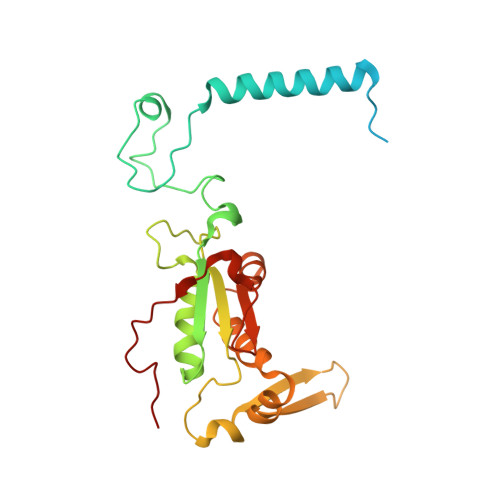
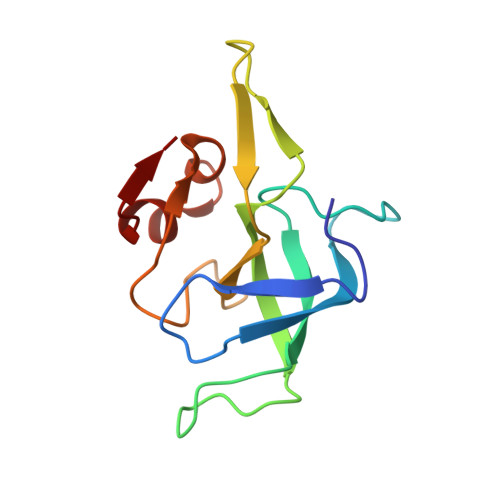
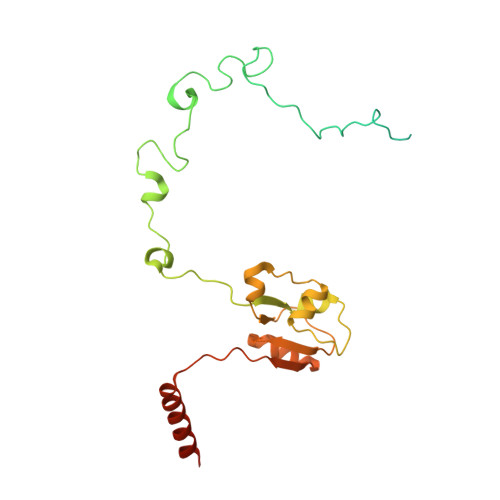
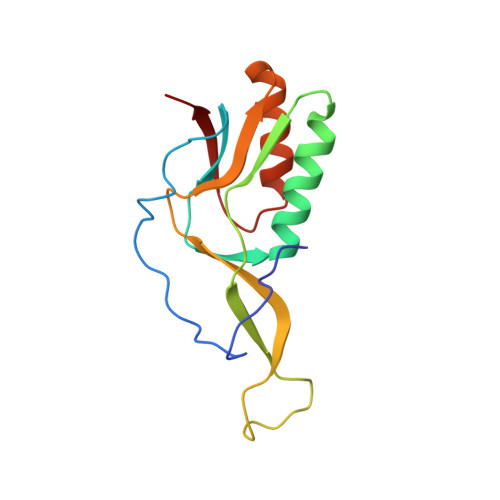
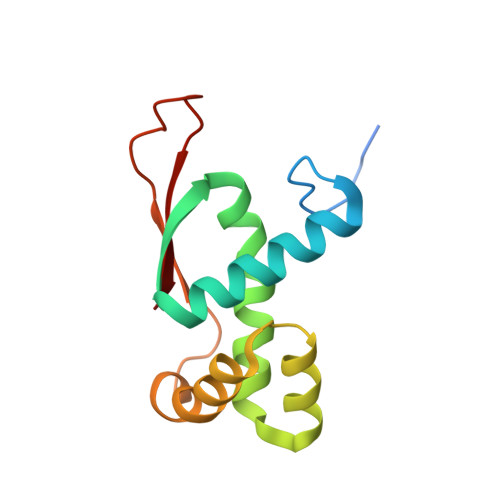
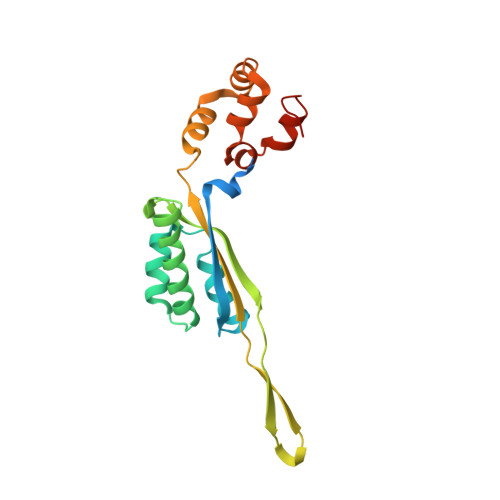
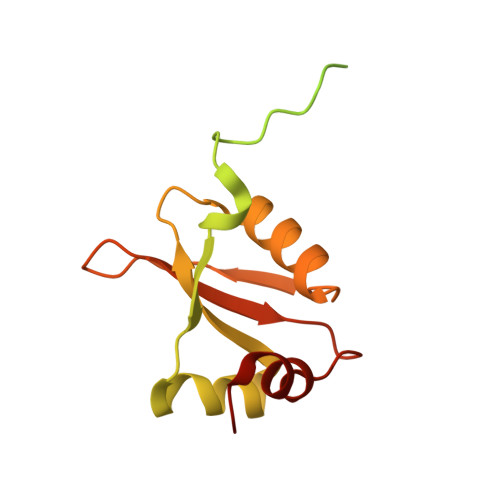
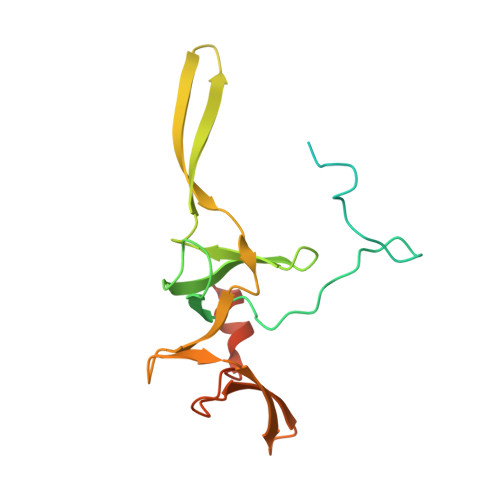
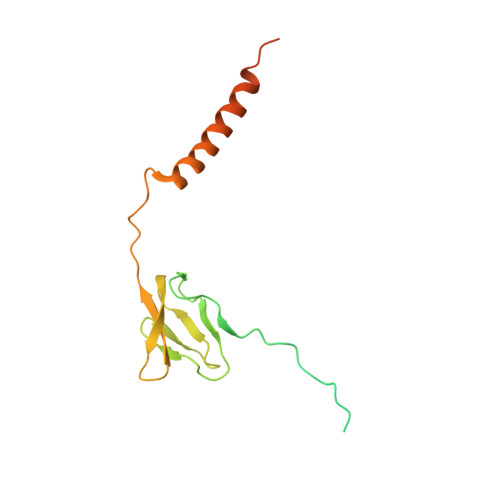
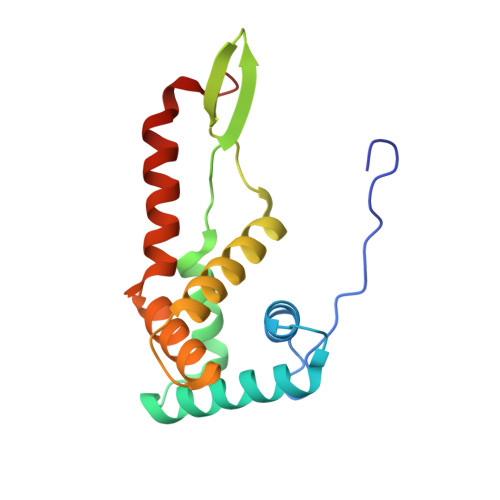
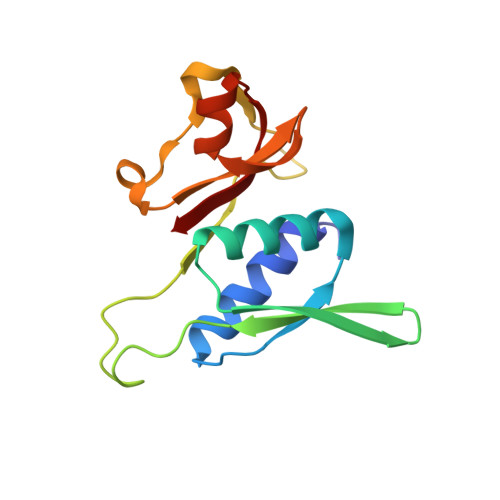
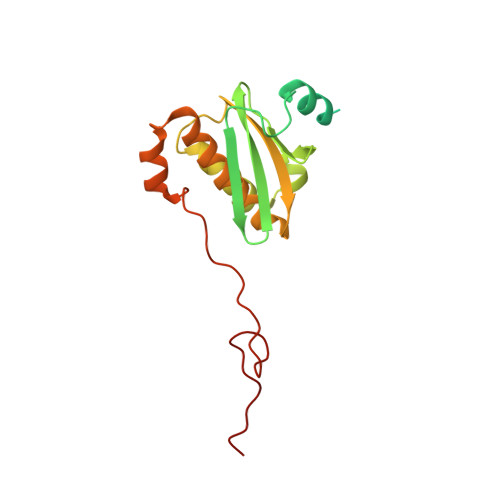
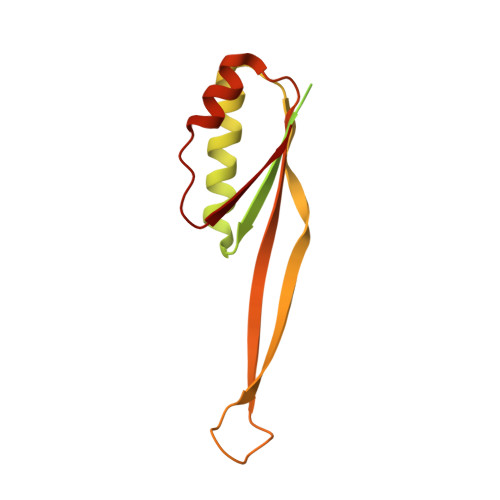
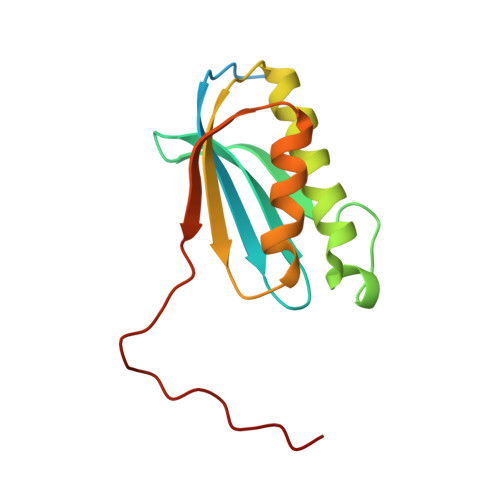
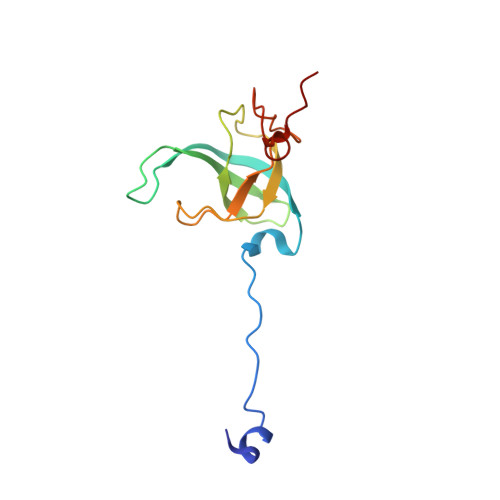
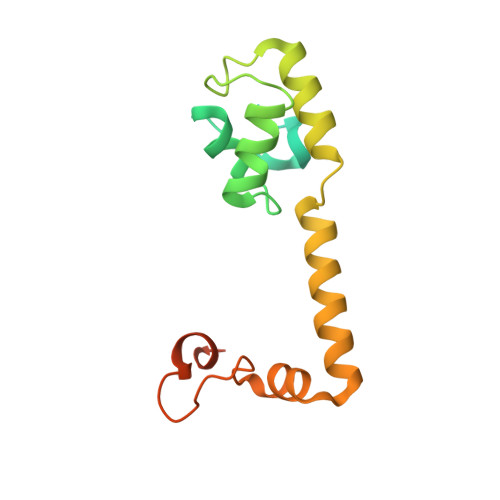
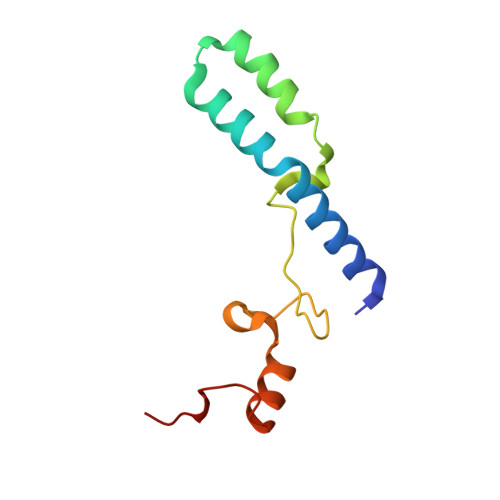
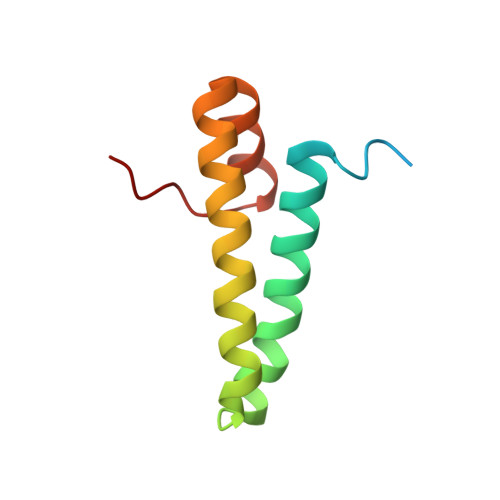
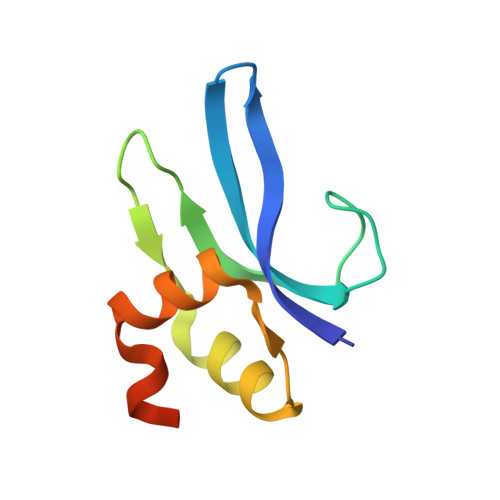
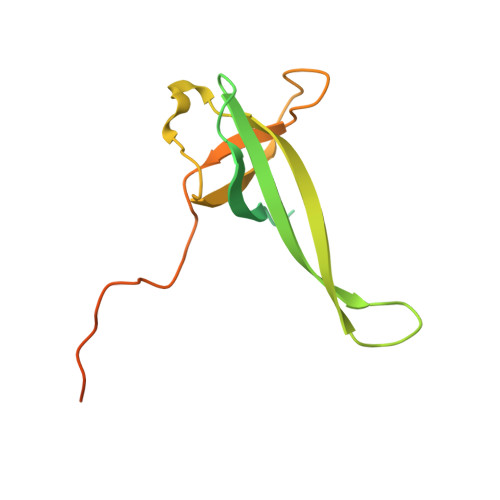
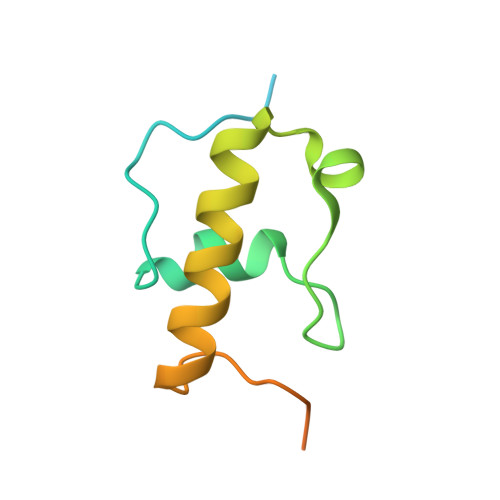
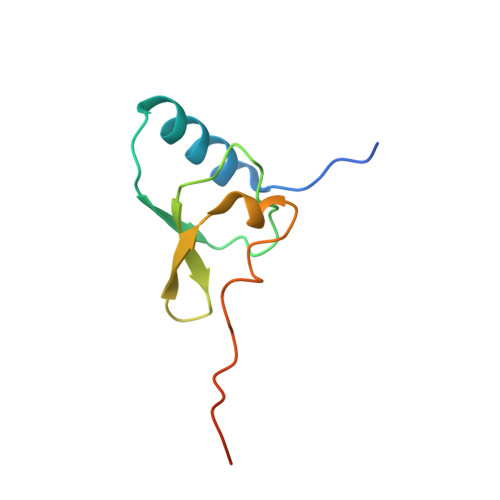
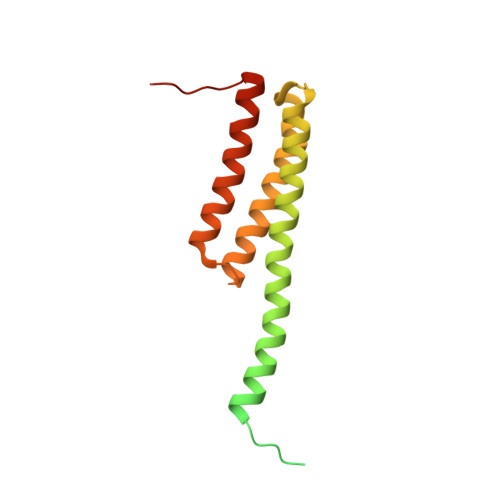
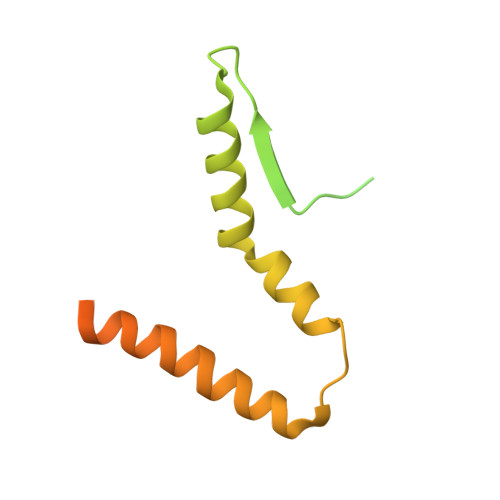
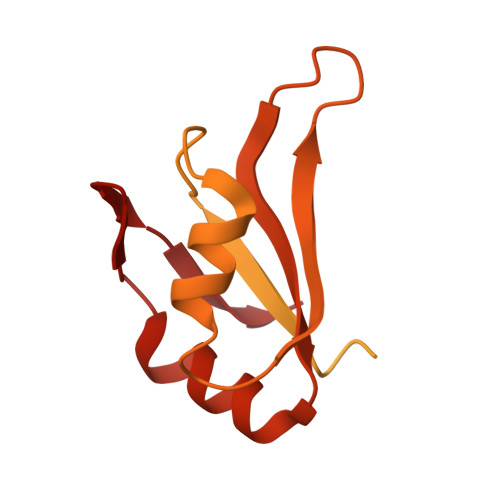
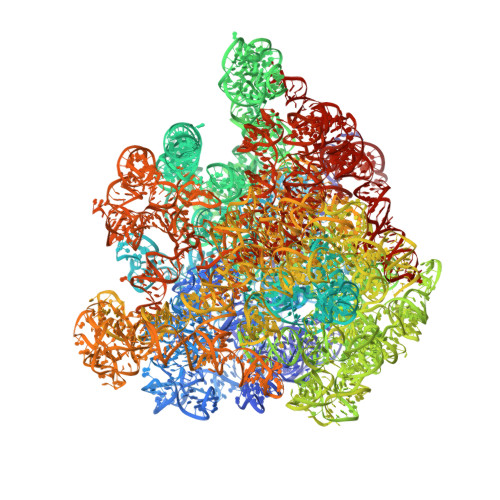
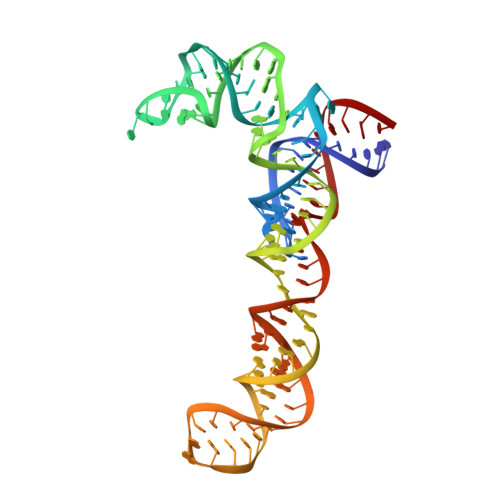
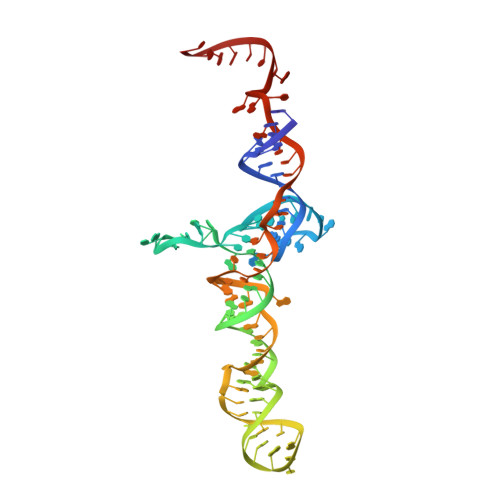
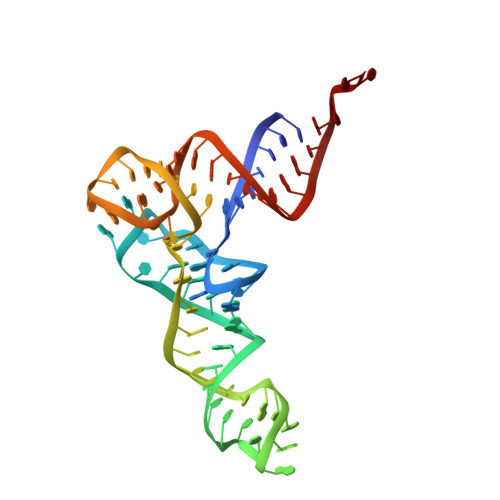
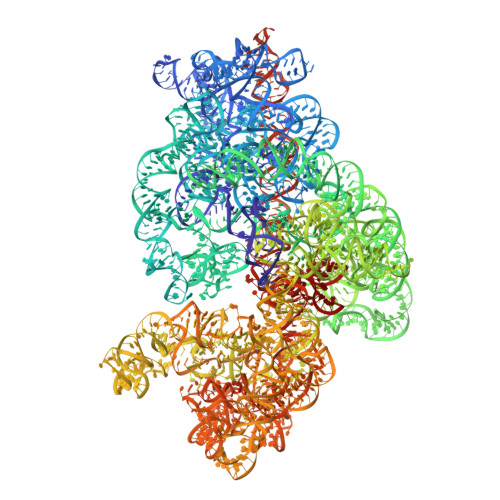
MG Query on MG
Download: Ideal Coordinates CCD File AAA [auth a]
AAA [auth a],
MAGNESIUM ION