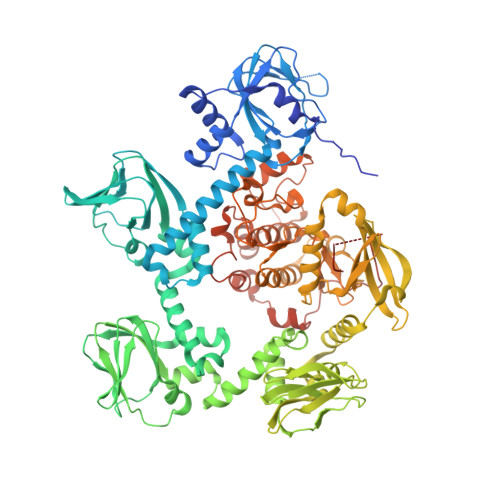
Structures of the cGMP-dependent protein kinase in malaria parasites reveal a unique structural relay mechanism for activation.
El Bakkouri, M., Kouidmi, I., Wernimont, A.K., Amani, M., Hutchinson, A., Loppnau, P., Kim, J.J., Flueck, C., Walker, J.R., Seitova, A., Senisterra, G., Kakihara, Y., Kim, C., Blackman, M.J., Calmettes, C., Baker, D.A., Hui, R.(2019) Proc Natl Acad Sci U S A 116: 14164-14173
- PubMed: 31239348 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1905558116
- Primary Citation Related Structures:
5DYK, 5DYL, 5DZC, 5E16 - PubMed Abstract:
The cyclic guanosine-3',5'-monophosphate (cGMP)-dependent protein kinase (PKG) was identified >25 y ago; however, efforts to obtain a structure of the entire PKG enzyme or catalytic domain from any species have failed. In malaria parasites, cooperative activation of PKG triggers crucial developmental transitions throughout the complex life cycle. We have determined the cGMP-free crystallographic structures of PKG from Plasmodium falciparum and Plasmodium vivax , revealing how key structural components, including an N-terminal autoinhibitory segment (AIS), four predicted cyclic nucleotide-binding domains (CNBs), and a kinase domain (KD), are arranged when the enzyme is inactive. The four CNBs and the KD are in a pentagonal configuration, with the AIS docked in the substrate site of the KD in a swapped-domain dimeric arrangement. We show that although the protein is predominantly a monomer (the dimer is unlikely to be representative of the physiological form), the binding of the AIS is necessary to keep Plasmodium PKG inactive. A major feature is a helix serving the dual role of the N-terminal helix of the KD as well as the capping helix of the neighboring CNB. A network of connecting helices between neighboring CNBs contributes to maintaining the kinase in its inactive conformation. We propose a scheme in which cooperative binding of cGMP, beginning at the CNB closest to the KD, transmits conformational changes around the pentagonal molecule in a structural relay mechanism, enabling PKG to orchestrate rapid, highly regulated developmental switches in response to dynamic modulation of cGMP levels in the parasite.
- Structural Genomics Consortium, University of Toronto, Toronto, ON M5G 1L7, Canada.
Organizational Affiliation: