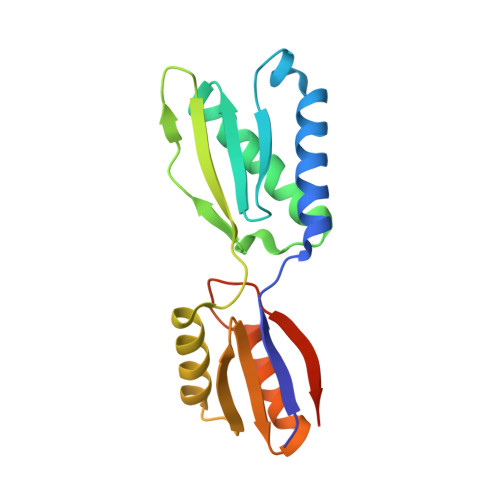
Crystal structure of the YajQ-family protein XC_3703 from Xanthomonas campestris pv. campestris
Zhao, Z., Wu, Z., Zhang, J.(2016) Acta Crystallogr F Struct Biol Commun 72: 720-725
- PubMed: 27599864 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S2053230X16013017
- Primary Citation Related Structures:
5B7W - PubMed Abstract:
As an important bacterial second messenger, bis-(3',5')-cyclic diguanylate (cyclic di-GMP or c-di-GMP) has been implicated in numerous biological activities, including biofilm formation, motility, survival and virulence. These processes are manipulated by the binding of c-di-GMP to its receptors. XC_3703 from the plant pathogen Xanthomonas campestris pv. campestris, which belongs to the YajQ family of proteins, has recently been identified as a potential c-di-GMP receptor. XC_3703, together with XC_2801, functions as a transcription factor activating virulence-related genes, which can be reversed by the binding of c-di-GMP to XC_3703. However, the structural basis of how c-di-GMP regulates XC_3703 remains elusive. In this study, the structure of XC_3703 was determined to 2.1 Å resolution using the molecular-replacement method. The structure of XC_3703 consists of two domains adopting the same topology, which is similar to that of the RNA-recognition motif (RRM). Arg65, which is conserved among the c-di-GMP-binding subfamily of the YajQ family of proteins, together with Phe80 in domain II, forms a putative c-di-GMP binding site.
- College of Biology, Pharmacy and Food Engineering, Shangluo University, Shangluo 726000, People's Republic of China.
Organizational Affiliation: