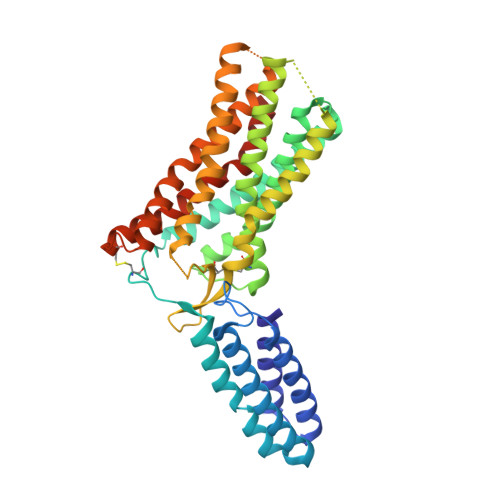
Structural Basis for Ligand Recognition and Functional Selectivity at Angiotensin Receptor.
Zhang, H., Unal, H., Desnoyer, R., Han, G.W., Patel, N., Katritch, V., Karnik, S.S., Cherezov, V., Stevens, R.C.(2015) J Biological Chem 290: 29127-29139
- PubMed: 26420482 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M115.689000
- Primary Citation Related Structures:
4ZUD - PubMed Abstract:
Angiotensin II type 1 receptor (AT1R) is the primary blood pressure regulator. AT1R blockers (ARBs) have been widely used in clinical settings as anti-hypertensive drugs and share a similar chemical scaffold, although even minor variations can lead to distinct therapeutic efficacies toward cardiovascular etiologies. The structural basis for AT1R modulation by different peptide and non-peptide ligands has remained elusive. Here, we report the crystal structure of the human AT1R in complex with an inverse agonist olmesartan (Benicar(TM)), a highly potent anti-hypertensive drug. Olmesartan is anchored to the receptor primarily by the residues Tyr-35(1.39), Trp-84(2.60), and Arg-167(ECL2), similar to the antagonist ZD7155, corroborating a common binding mode of different ARBs. Using docking simulations and site-directed mutagenesis, we identified specific interactions between AT1R and different ARBs, including olmesartan derivatives with inverse agonist, neutral antagonist, or agonist activities. We further observed that the mutation N111(3.35)A in the putative sodium-binding site affects binding of the endogenous peptide agonist angiotensin II but not the β-arrestin-biased peptide TRV120027.
- From the Departments of Biological Sciences and Chemistry, Bridge Institute, University of Southern California, Los Angeles, California 90089 and.
Organizational Affiliation: