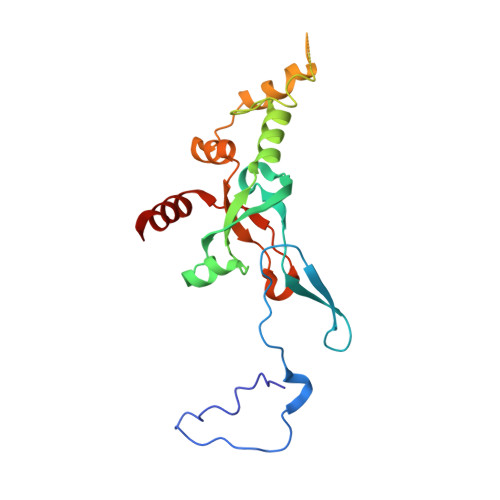
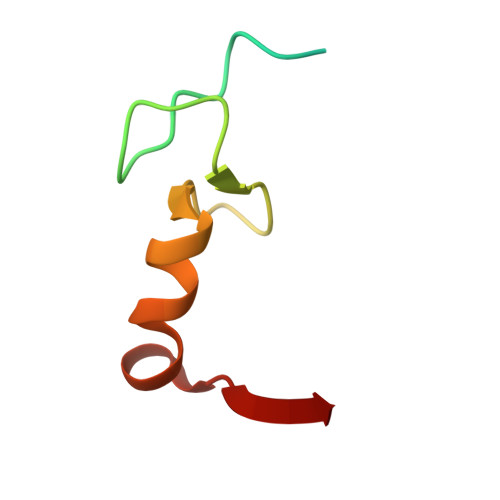
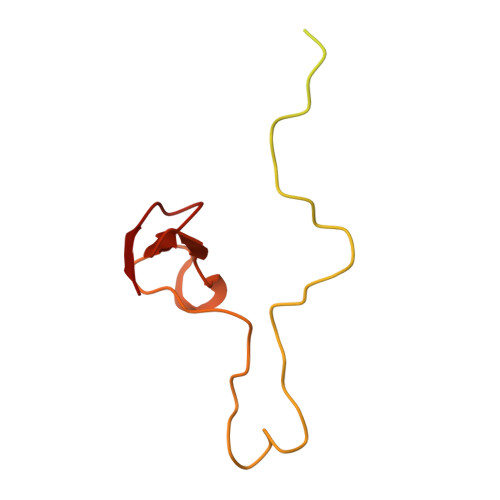
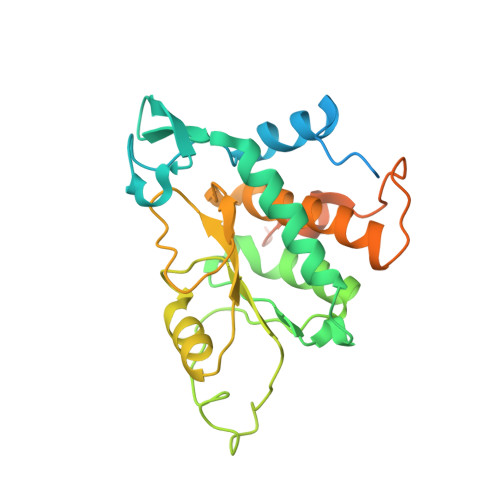
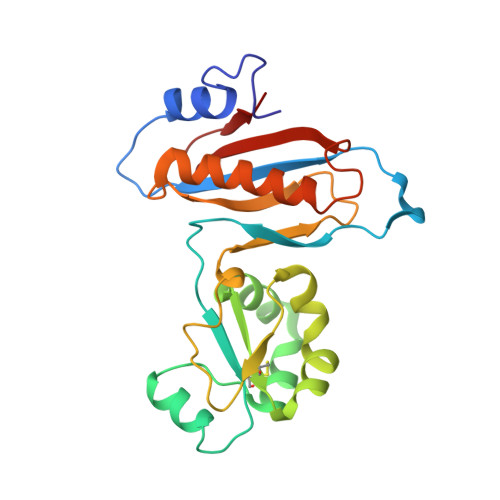
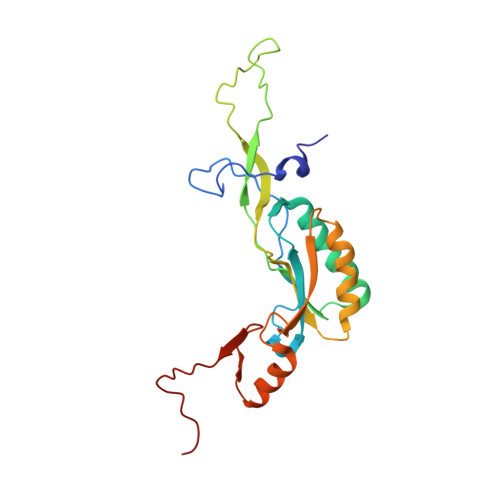
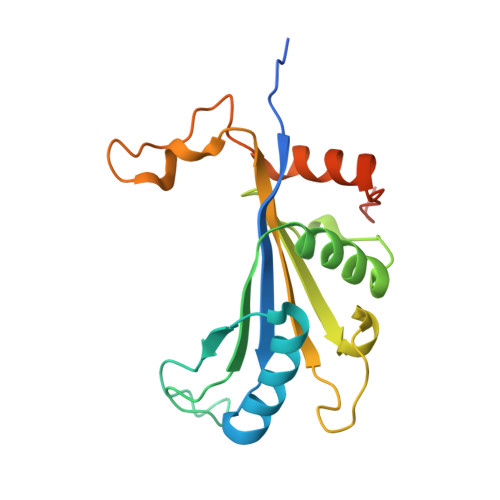
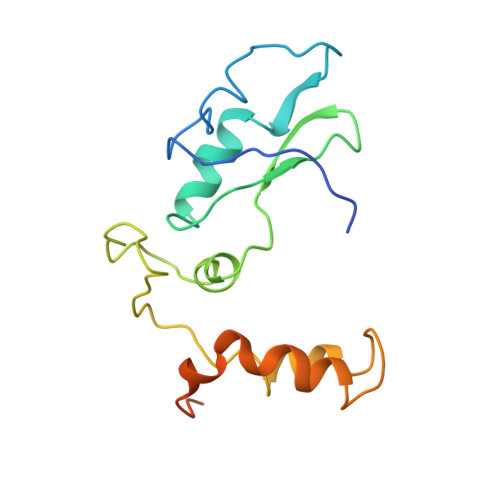
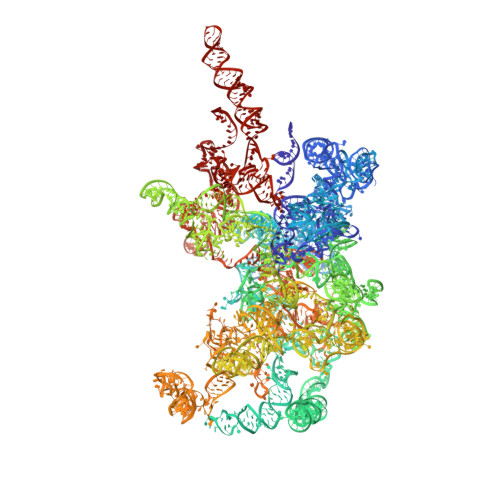
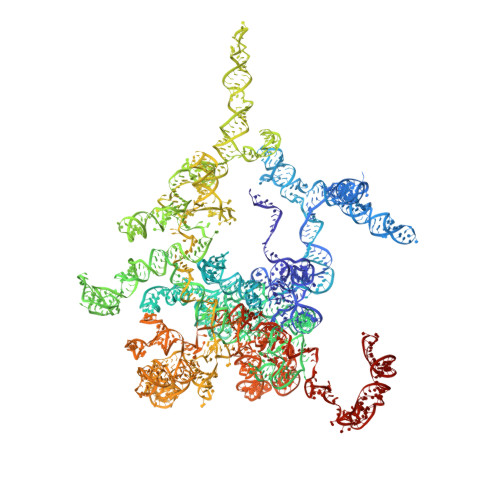
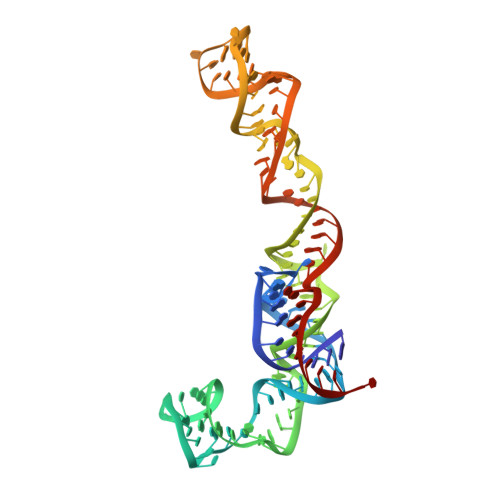
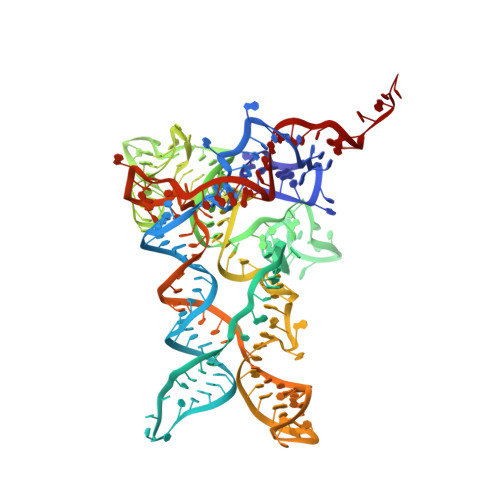
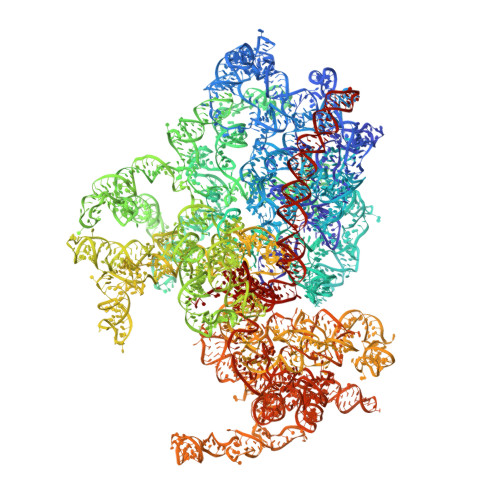
High-Resolution Cryo-Electron Microscopy Structure of the Trypanosoma Brucei Ribosome.
Hashem, Y., Des Georges, A., Fu, J., Buss, S.N., Jossinet, F., Jobe, A., Zhang, Q., Liao, H.Y., Grassucci, R.A., Bajaj, C., Westhof, E., Madison-Antenucci, S., Frank, J.(2013) Nature 494: 385
- PubMed: 23395961 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nature11872
- Primary Citation Related Structures:
4V8M - PubMed Abstract:
Ribosomes, the protein factories of living cells, translate genetic information carried by messenger RNAs into proteins, and are thus involved in virtually all aspects of cellular development and maintenance. The few available structures of the eukaryotic ribosome reveal that it is more complex than its prokaryotic counterpart, owing mainly to the presence of eukaryote-specific ribosomal proteins and additional ribosomal RNA insertions, called expansion segments. The structures also differ among species, partly in the size and arrangement of these expansion segments. Such differences are extreme in kinetoplastids, unicellular eukaryotic parasites often infectious to humans. Here we present a high-resolution cryo-electron microscopy structure of the ribosome of Trypanosoma brucei, the parasite that is transmitted by the tsetse fly and that causes African sleeping sickness. The atomic model reveals the unique features of this ribosome, characterized mainly by the presence of unusually large expansion segments and ribosomal-protein extensions leading to the formation of four additional inter-subunit bridges. We also find additional rRNA insertions, including one large rRNA domain that is not found in other eukaryotes. Furthermore, the structure reveals the five cleavage sites of the kinetoplastid large ribosomal subunit (LSU) rRNA chain, which is known to be cleaved uniquely into six pieces, and suggests that the cleavage is important for the maintenance of the T. brucei ribosome in the observed structure. We discuss several possible implications of the large rRNA expansion segments for the translation-regulation process. The structure could serve as a basis for future experiments aimed at understanding the functional importance of these kinetoplastid-specific ribosomal features in protein-translation regulation, an essential step towards finding effective and safe kinetoplastid-specific drugs.
- Howard Hughes Medical Institute, Department of Biochemistry and Molecular Biophysics, Columbia University, New York, New York 10032, USA.
Organizational Affiliation: