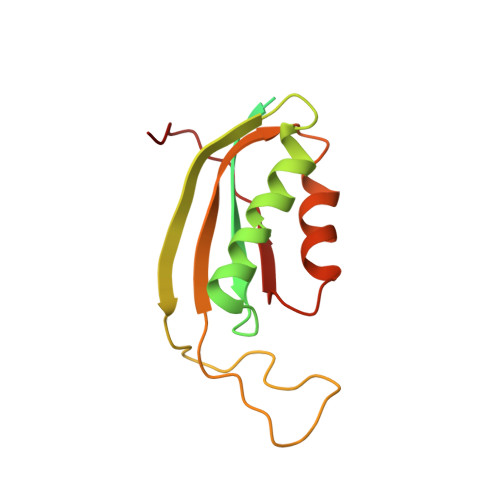
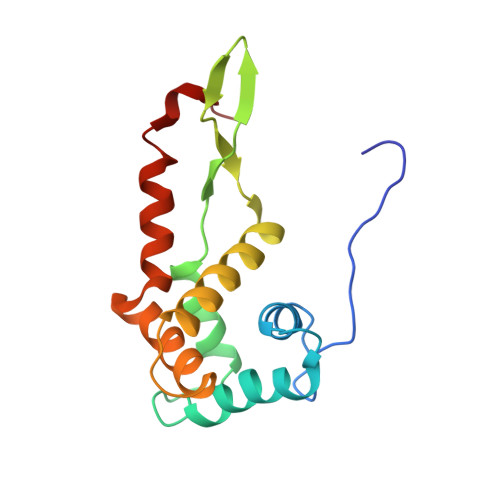
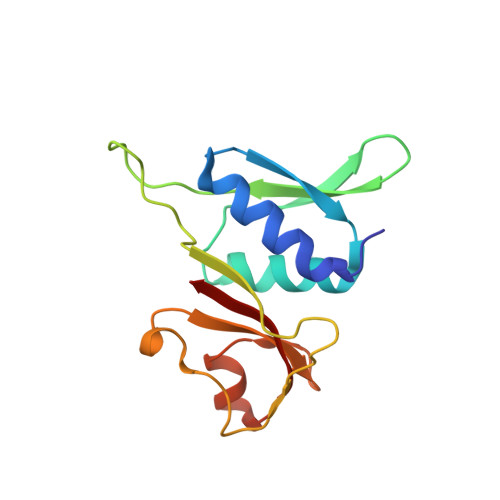
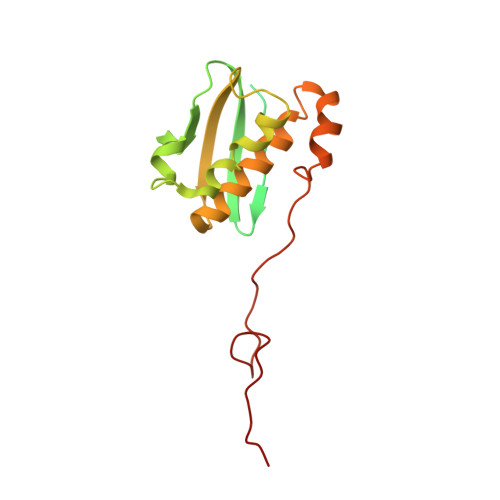
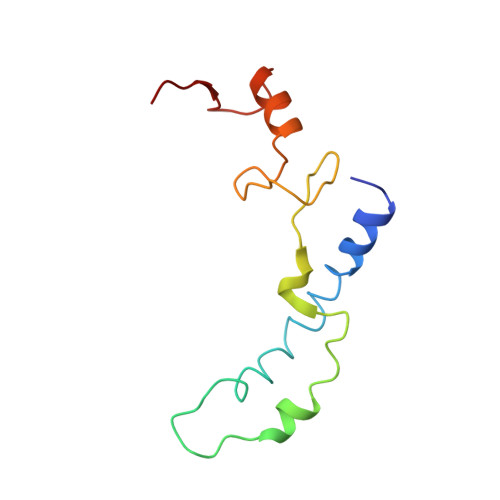
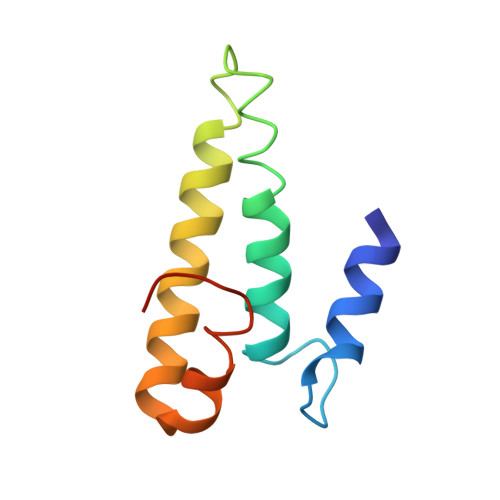
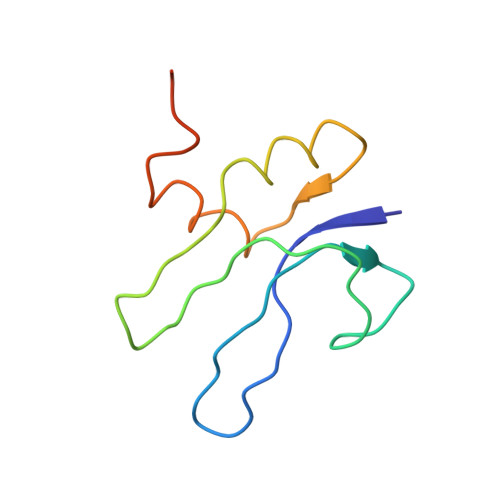
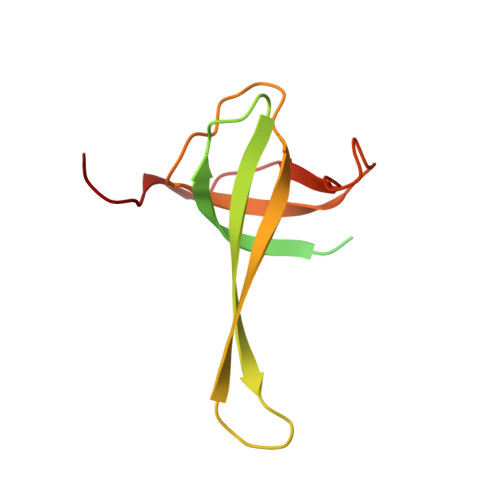
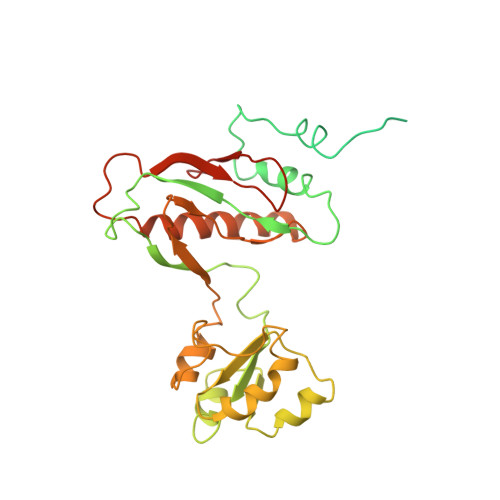
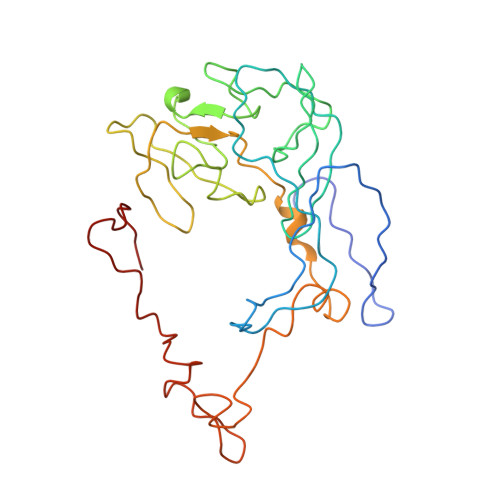
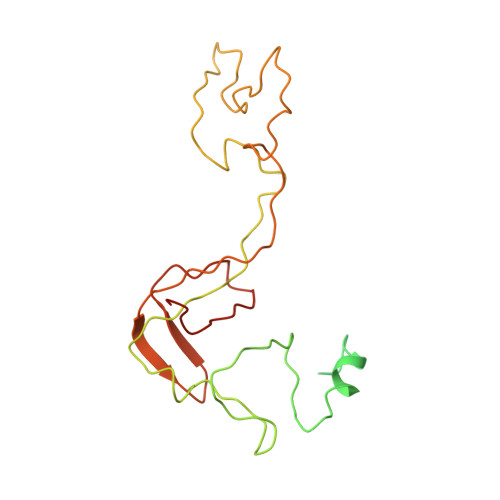
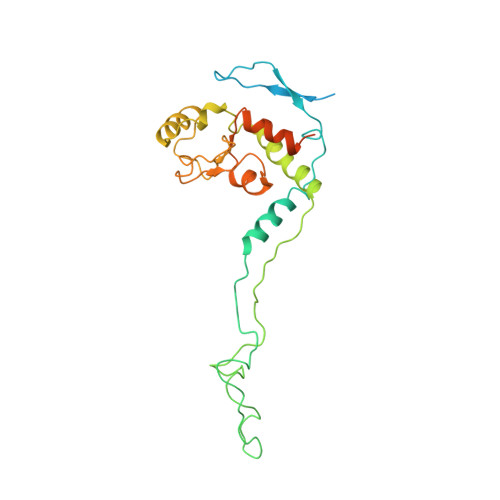
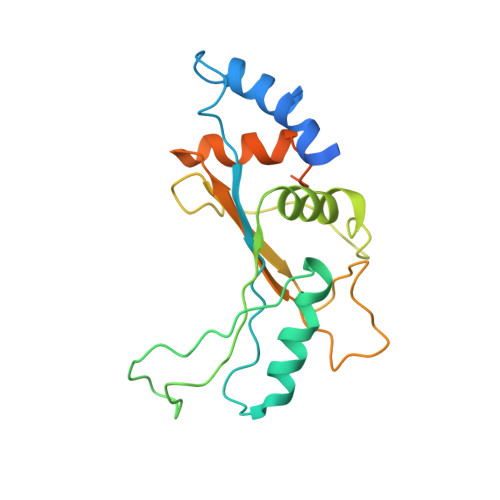
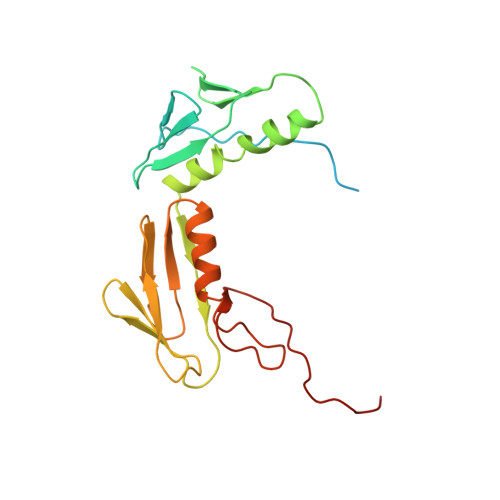
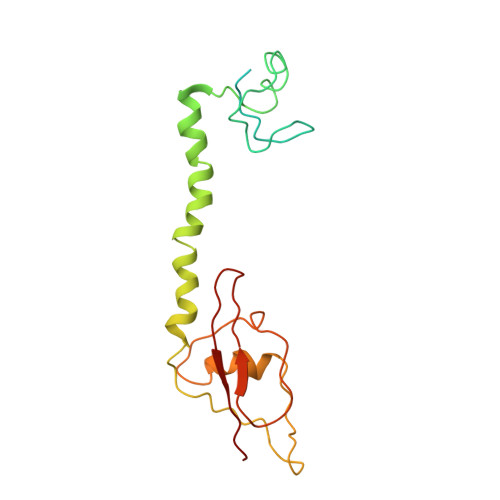
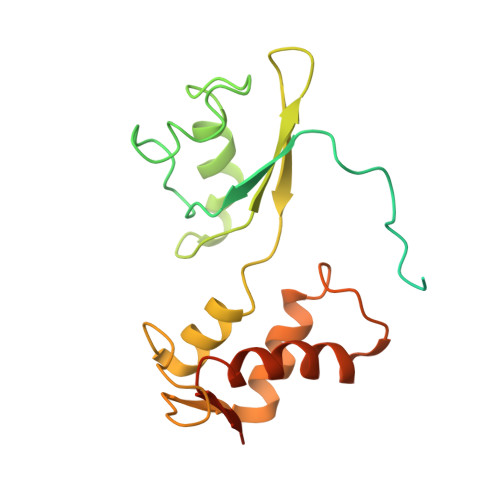
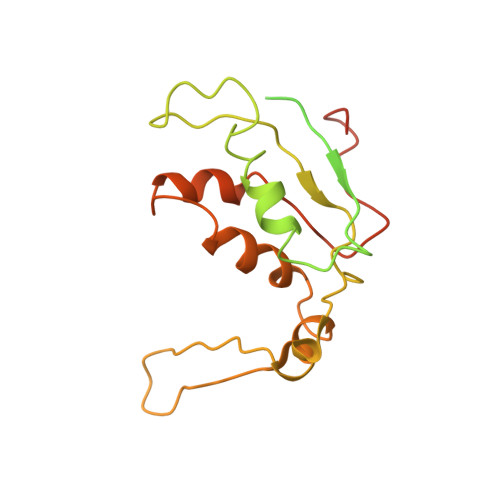
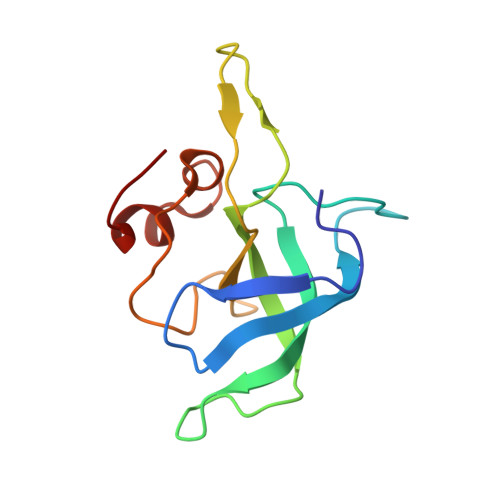
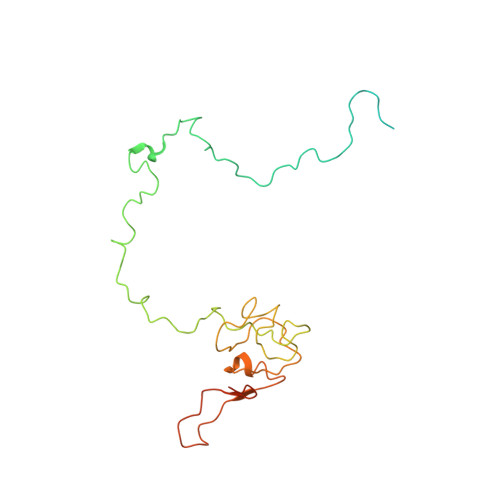
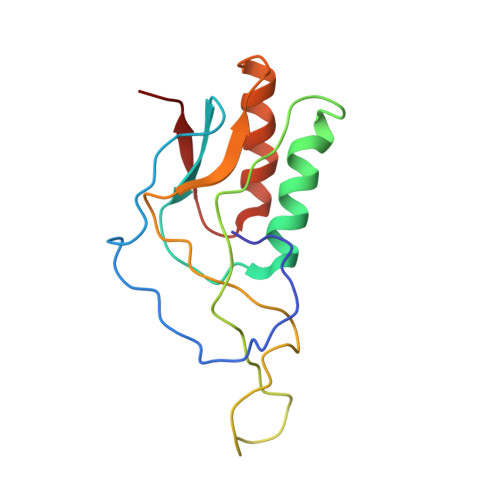
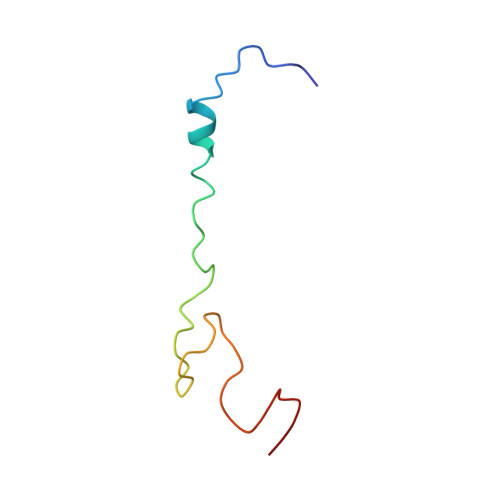
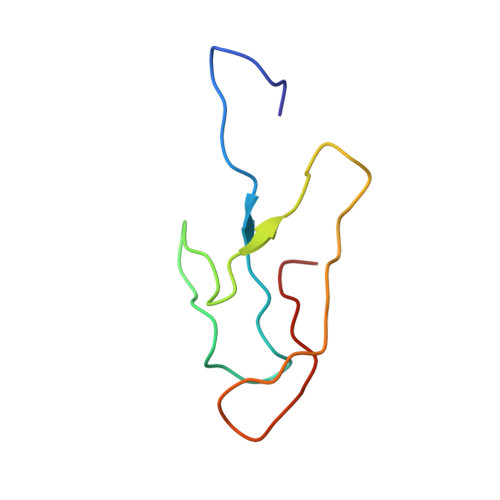
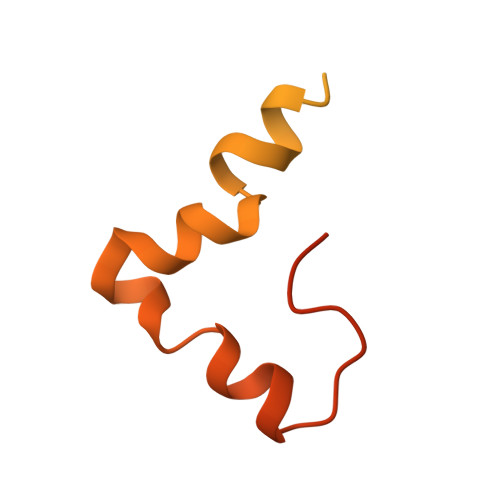
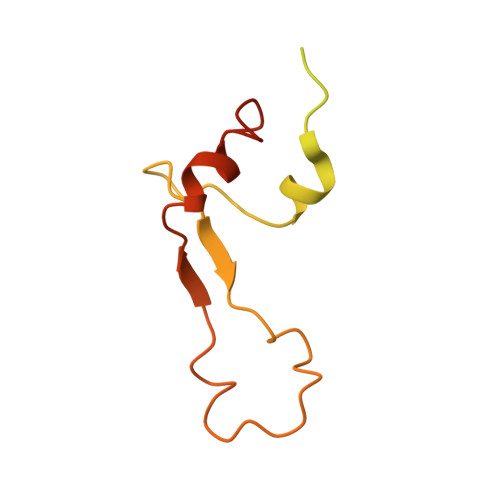
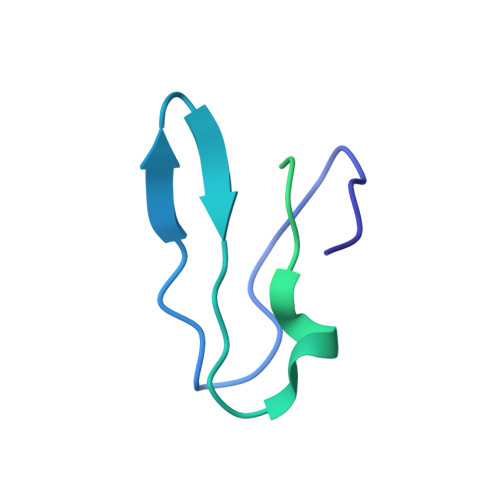
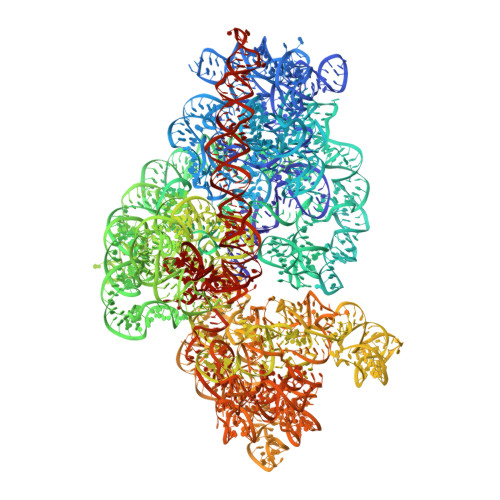
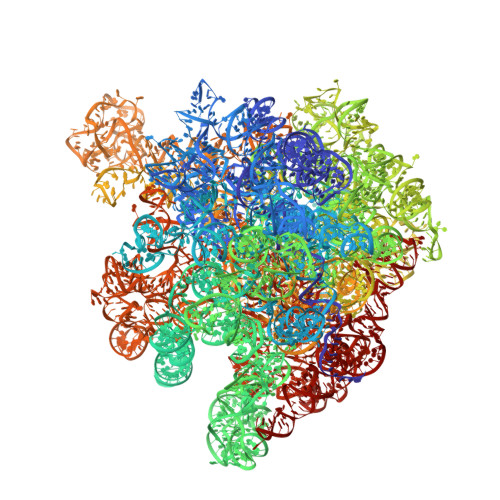
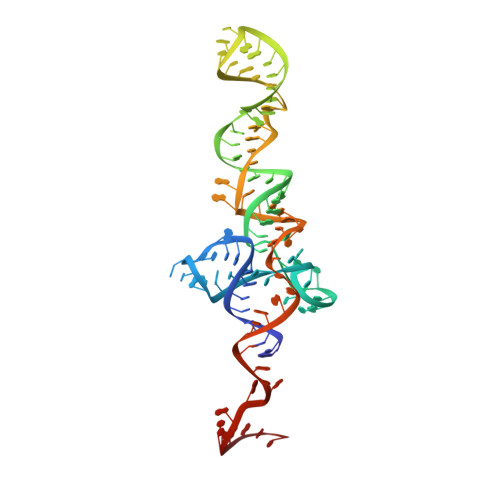
Cryo-EM study of the spinach chloroplast ribosome reveals the structural and functional roles of plastid-specific ribosomal proteins
Sharma, M.R., Wilson, D.N., Datta, P.P., Barat, C., Schluenzen, F., Fucini, P., Agrawal, R.K.(2007) Proc Natl Acad Sci U S A 104: 19315-19320
- PubMed: 18042701 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0709856104
- Primary Citation Related Structures:
4V61 - PubMed Abstract:
Protein synthesis in the chloroplast is carried out by chloroplast ribosomes (chloro-ribosome) and regulated in a light-dependent manner. Chloroplast or plastid ribosomal proteins (PRPs) generally are larger than their bacterial counterparts, and chloro-ribosomes contain additional plastid-specific ribosomal proteins (PSRPs); however, it is unclear to what extent these proteins play structural or regulatory roles during translation. We have obtained a three-dimensional cryo-EM map of the spinach 70S chloro-ribosome, revealing the overall structural organization to be similar to bacterial ribosomes. Fitting of the conserved portions of the x-ray crystallographic structure of the bacterial 70S ribosome into our cryo-EM map of the chloro-ribosome reveals the positions of PRP extensions and the locations of the PSRPs. Surprisingly, PSRP1 binds in the decoding region of the small (30S) ribosomal subunit, in a manner that would preclude the binding of messenger and transfer RNAs to the ribosome, suggesting that PSRP1 is a translation factor rather than a ribosomal protein. PSRP2 and PSRP3 appear to structurally compensate for missing segments of the 16S rRNA within the 30S subunit, whereas PSRP4 occupies a position buried within the head of the 30S subunit. One of the two PSRPs in the large (50S) ribosomal subunit lies near the tRNA exit site. Furthermore, we find a mass of density corresponding to chloro-ribosome recycling factor; domain II of this factor appears to interact with the flexible C-terminal domain of PSRP1. Our study provides evolutionary insights into the structural and functional roles that the PSRPs play during protein synthesis in chloroplasts.
- Laboratory of Structural Pathology, Division of Molecular Medicine, Wadsworth Center, New York State Department of Health, Empire State Plaza, Albany, NY 12201-0509, USA.
Organizational Affiliation: