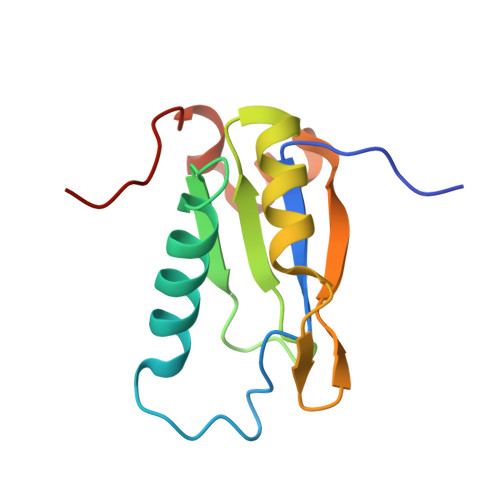
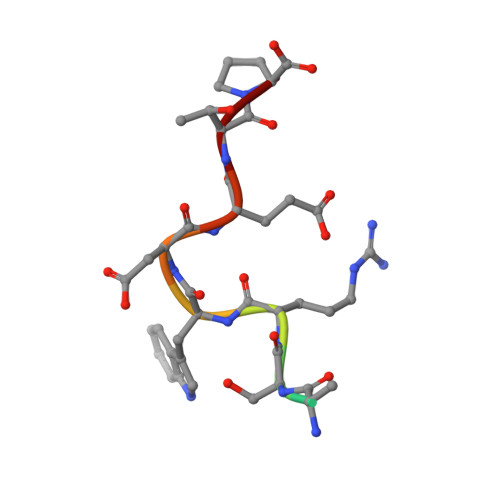
Cancer-relevant splicing factor CAPER alpha engages the essential splicing factor SF3b155 in a specific ternary complex.
Loerch, S., Maucuer, A., Manceau, V., Green, M.R., Kielkopf, C.L.(2014) J Biological Chem 289: 17325-17337
- PubMed: 24795046 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M114.558825
- Primary Citation Related Structures:
4OZ0, 4OZ1 - PubMed Abstract:
U2AF homology motifs (UHMs) mediate protein-protein interactions with U2AF ligand motifs (ULMs) of pre-mRNA splicing factors. The UHM-containing alternative splicing factor CAPERα regulates splicing of tumor-promoting VEGF isoforms, yet the molecular target of the CAPERα UHM is unknown. Here we present structures of the CAPERα UHM bound to a representative SF3b155 ULM at 1.7 Å resolution and, for comparison, in the absence of ligand at 2.2 Å resolution. The prototypical UHM/ULM interactions authenticate CAPERα as a bona fide member of the UHM family of proteins. We identify SF3b155 as the relevant ULM-containing partner of full-length CAPERα in human cell extracts. Isothermal titration calorimetry comparisons of the purified CAPERα UHM binding known ULM-containing proteins demonstrate that high affinity interactions depend on the presence of an intact, intrinsically unstructured SF3b155 domain containing seven ULM-like motifs. The interplay among bound CAPERα molecules gives rise to the appearance of two high affinity sites in the SF3b155 ULM-containing domain. In conjunction with the previously identified, UHM/ULM-mediated complexes of U2AF(65) and SPF45 with SF3b155, this work demonstrates the capacity of SF3b155 to offer a platform for coordinated recruitment of UHM-containing splicing factors.
- From the Department of Biochemistry and Biophysics, University of Rochester School of Medicine and Dentistry, Rochester, New York 14642 and.
Organizational Affiliation: