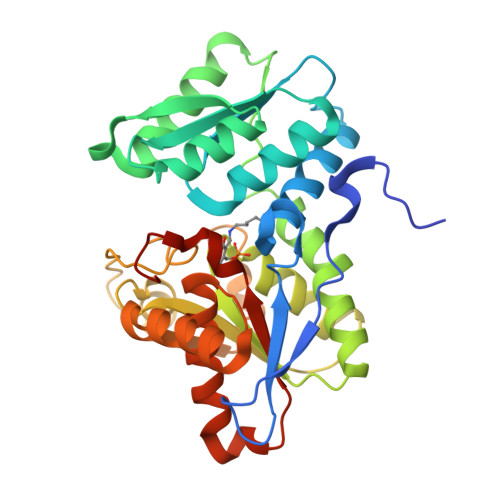
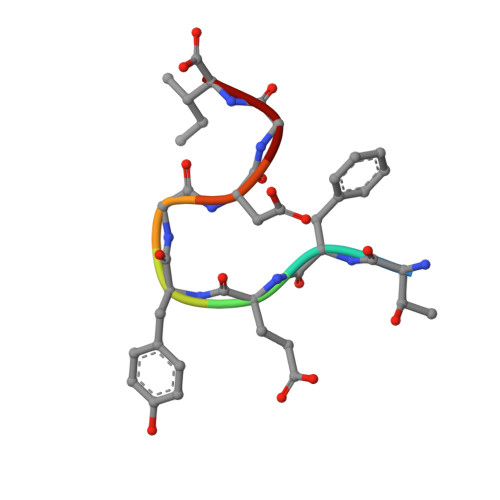
Crystal structure of O-acetylserine sulfhydrylase from Haemophilus influenzae in complex with high affinity inhibitory peptide from serine acetyl transferase of Salmonella Typhimurium at 2.0 A
Ekka, M.K., Kaushik, A., Singh, A.K., Kumaran, S.To be published.