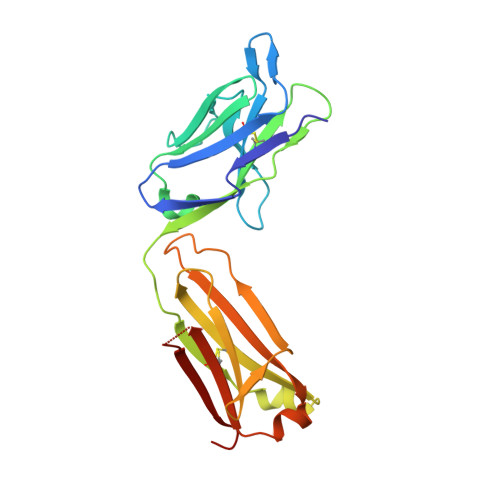
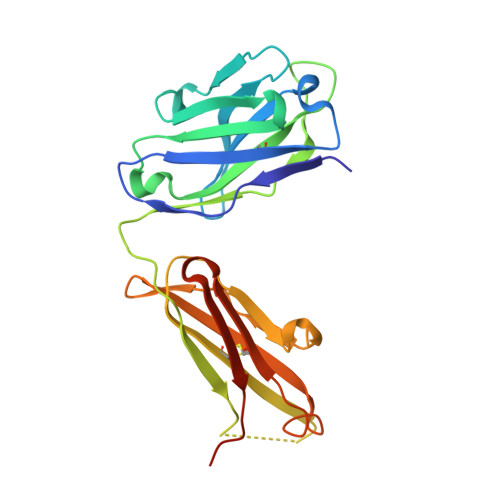
Adaptive Mutations Alter Antibody Structure and Dynamics during Affinity Maturation.
Adhikary, R., Yu, W., Oda, M., Walker, R.C., Chen, T., Stanfield, R.L., Wilson, I.A., Zimmermann, J., Romesberg, F.E.(2015) Biochemistry 54: 2085-2093
- PubMed: 25756188 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/bi501417q
- Primary Citation Related Structures:
4NJ9, 4NJA - PubMed Abstract:
While adaptive mutations can bestow new functions on proteins via the introduction or optimization of reactive centers, or other structural changes, a role for the optimization of protein dynamics also seems likely but has been more difficult to evaluate. Antibody (Ab) affinity maturation is an example of adaptive evolution wherein the adaptive mutations may be identified and Abs may be raised to specific targets that facilitate the characterization of protein dynamics. Here, we report the characterization of three affinity matured Abs that evolved from a common germline precursor to bind the chromophoric antigen (Ag), 8-methoxypyrene-1,3,6-trisulfonate (MPTS). In addition to characterizing the sequence, molecular recognition, and structure of each Ab, we characterized the dynamics of each complex by determining their mechanical response to an applied force via three-pulse photon echo peak shift (3PEPS) spectroscopy and deconvoluting the response into elastic, anelastic, and plastic components. We find that for one Ab, affinity maturation was accomplished via the introduction of a single functional group that mediates a direct contact with MPTS and results in a complex with little anelasticity or plasticity. In the other two cases, more mutations were introduced but none directly contact MPTS, and while their effects on structure are subtle, their effects on anelasticity and plasticity are significant, with the level of plasticity correlated with specificity, suggesting that the optimization of protein dynamics may have contributed to affinity maturation. A similar optimization of structure and dynamics may contribute to the evolution of other proteins.
- §Graduate School of Life and Environmental Sciences, Kyoto Prefectural University, 1-5, Hangi-cho, Shimogamo, Sakyo-ku, Kyoto 606-8522, Japan.
Organizational Affiliation: