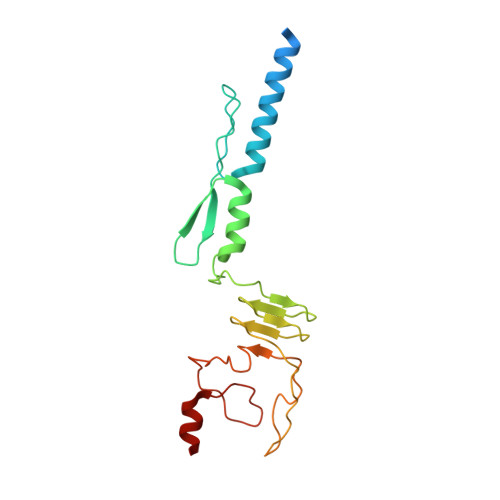
Structure of a Burkholderia pseudomallei trimeric autotransporter adhesin head.
Edwards, T.E., Phan, I., Abendroth, J., Dieterich, S.H., Masoudi, A., Guo, W., Hewitt, S.N., Kelley, A., Leibly, D., Brittnacher, M.J., Staker, B.L., Miller, S.I., Van Voorhis, W.C., Myler, P.J., Stewart, L.J.(2010) PLoS One 5: 12803-12811
- PubMed: 20862217 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0012803
- Primary Citation Related Structures:
3LA9, 3LAA - PubMed Abstract:
Pathogenic bacteria adhere to the host cell surface using a family of outer membrane proteins called Trimeric Autotransporter Adhesins (TAAs). Although TAAs are highly divergent in sequence and domain structure, they are all conceptually comprised of a C-terminal membrane anchoring domain and an N-terminal passenger domain. Passenger domains consist of a secretion sequence, a head region that facilitates binding to the host cell surface, and a stalk region. Pathogenic species of Burkholderia contain an overabundance of TAAs, some of which have been shown to elicit an immune response in the host. To understand the structural basis for host cell adhesion, we solved a 1.35 A resolution crystal structure of a BpaA TAA head domain from Burkholderia pseudomallei, the pathogen that causes melioidosis. The structure reveals a novel fold of an intricately intertwined trimer. The BpaA head is composed of structural elements that have been observed in other TAA head structures as well as several elements of previously unknown structure predicted from low sequence homology between TAAs. These elements are typically up to 40 amino acids long and are not domains, but rather modular structural elements that may be duplicated or omitted through evolution, creating molecular diversity among TAAs. The modular nature of BpaA, as demonstrated by its head domain crystal structure, and of TAAs in general provides insights into evolution of pathogen-host adhesion and may provide an avenue for diagnostics.
- Seattle Structural Genomics Center for Infectious Disease, Seattle, Washington, USA. tedwards@embios.com
Organizational Affiliation: