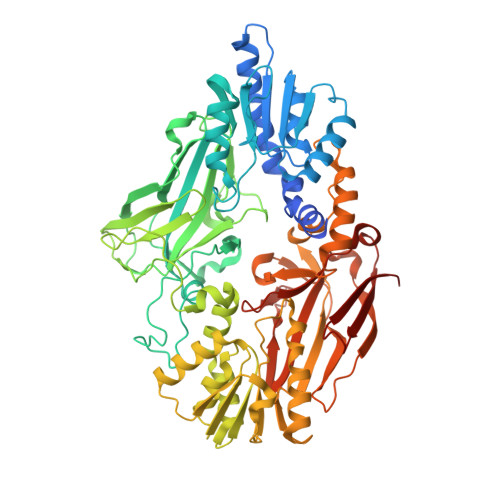
Protein arginine methyltransferase 7 has a novel homodimer-like structure formed by tandem repeats
Hasegawa, M., Toma-fukai, S., Kim, J.D., Fukamizu, A., Shimizu, T.(2014) FEBS Lett 588: 1942-1948
- PubMed: 24726727 Search on PubMed
- DOI: https://doi.org/10.1016/j.febslet.2014.03.053
- Primary Citation Related Structures:
3WST, 3X0D - PubMed Abstract:
Protein arginine methyltransferase 7 (PRMT7) is a member of a family of enzymes that catalyze the transfer of methyl groups from S-adenosyl-l-methionine to nitrogen atoms on arginine residues. Here, we describe the crystal structure of Caenorhabditis elegans PRMT7 in complex with its reaction product S-adenosyl-L-homocysteine. The structural data indicated that PRMT7 harbors two tandem repeated PRMT core domains that form a novel homodimer-like structure. S-adenosyl-L-homocysteine bound to the N-terminal catalytic site only; the C-terminal catalytic site is occupied by a loop that inhibits cofactor binding. Mutagenesis demonstrated that only the N-terminal catalytic site of PRMT7 is responsible for cofactor binding.
- Graduate School of Pharmaceutical Sciences, The University of Tokyo, Hongo, Bunkyo-ku, Tokyo 113-0033, Japan.
Organizational Affiliation: