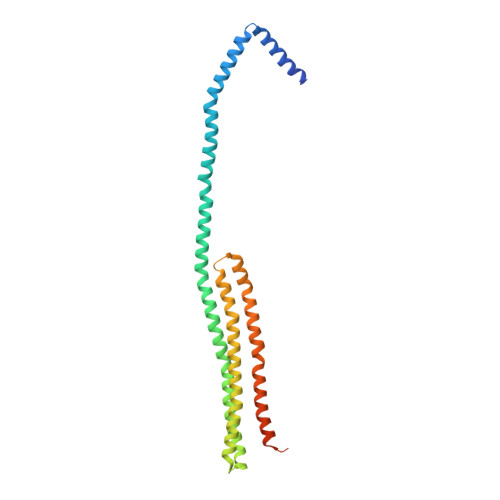
The Structure of Dimeric Apolipoprotein A-IV and Its Mechanism of Self-Association.
Deng, X., Morris, J., Dressmen, J., Tubb, M.R., Tso, P., Jerome, W.G., Davidson, W.S., Thompson, T.B.(2012) Structure 20: 767-779
- PubMed: 22579246 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2012.02.020
- Primary Citation Related Structures:
3S84 - PubMed Abstract:
Apolipoproteins are key structural elements of lipoproteins and critical mediators of lipid metabolism. Their detergent-like properties allow them to emulsify lipid or exist in a soluble lipid-free form in various states of self-association. Unfortunately, these traits have hampered high-resolution structural studies needed to understand the biogenesis of cardioprotective high-density lipoproteins (HDLs). We derived a crystal structure of the core domain of human apolipoprotein (apo)A-IV, an HDL component and important mediator of lipid absorption. The structure at 2.4 Å depicts two linearly connected 4-helix bundles participating in a helix swapping arrangement that offers a clear explanation for how the protein self-associates as well as clues to the structure of its monomeric form. This also provides a logical basis for antiparallel arrangements recently described for lipid-containing particles. Furthermore, we propose a "swinging door" model for apoA-IV lipid association.
- Department of Molecular Genetics, Biochemistry, and Microbiology, College of Medicine, University of Cincinnati, OH 45267, USA.
Organizational Affiliation: