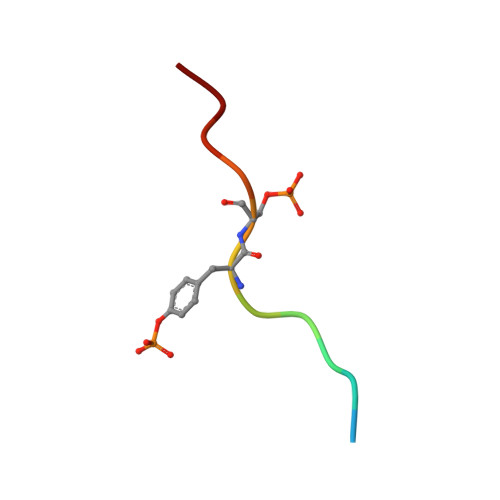
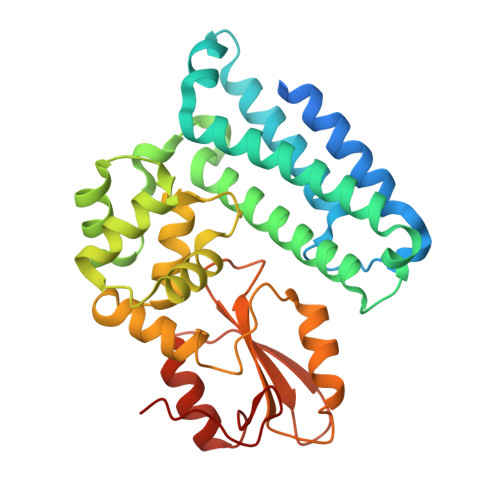
Additional serine/threonine phosphorylation reduces binding affinity but preserves interface topography of substrate proteins to the c-Cbl TKB domain
Sun, Q., Jackson, R.A., Ng, C., Guy, G.R., Sivaraman, J.(2010) PLoS One 5: e12819-e12819
- PubMed: 20877636
- DOI: https://doi.org/10.1371/journal.pone.0012819
- Primary Citation of Related Structures:
3OB1, 3OB2 - PubMed Abstract:
The E3-ubiquitin ligase, c-Cbl, is a multi-functional scaffolding protein that plays a pivotal role in controlling cell phenotype. As part of the ubiquitination and downregulation process, c-Cbl recognizes targets, such as tyrosine kinases and the Sprouty proteins, by binding to a conserved (NX/R)pY(S/T)XXP motif via its uniquely embedded SH2 domain (TKB domain). We previously outlined the mode of binding between the TKB domain and various substrate peptide motifs, including epidermal growth factor receptor (EGFR) and Sprouty2 (Spry2), and demonstrated that an intrapetidyl hydrogen bond forms between the (pY-1) arginine or (pY-2) asparagine and the phosphorylated tyrosine, which is crucial for binding. Recent reports demonstrated that, under certain types of stimulation, the serine/threonine residues at the pY+1 and/or pY+2 positions within this recognition motif of EGFR and Sprouty2 may be endogenously phosphorylated. Using structural and binding studies, we sought to determine whether this additional phosphorylation could affect the binding of the TKB domain to these peptides and consequently, whether the type of stimulation can dictate the degree to which substrates bind to c-Cbl. Here, we show that additional phosphorylation significantly reduces the binding affinity between the TKB domain and its target proteins, EGFR and Sprouty2, as compared to peptides bearing a single tyrosine phosphorylation. The crystal structure indicates that this is accomplished with minimal changes to the essential intrapeptidyl bond and that the reduced strength of the interaction is due to the charge repulsion between c-Cbl and the additional phosphate group. This obvious reduction in binding affinity, however, indicates that Cbl's interactions with its TKB-centered binding partners may be more favorable in the absence of Ser/Thr phosphorylation, which is stimulation and context specific in vivo. These results demonstrate the importance of understanding the environment in which certain residues are phosphorylated, and the necessity of including this in structural investigations.
- Department of Biological Sciences, National University of Singapore, Singapore, Singapore.
Organizational Affiliation: