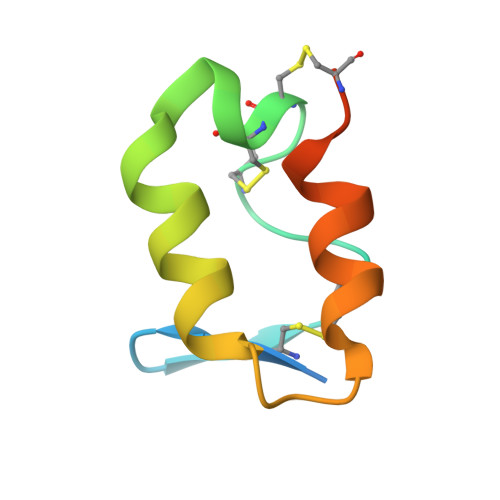
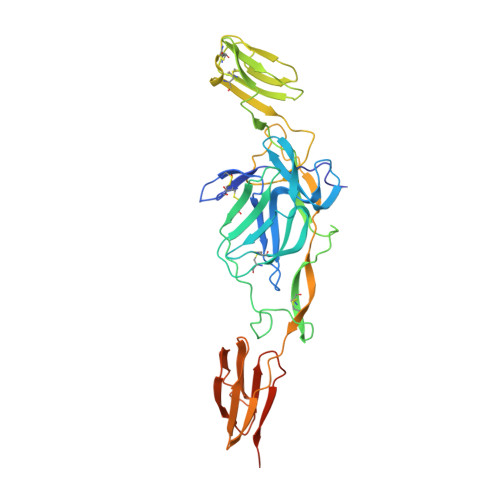
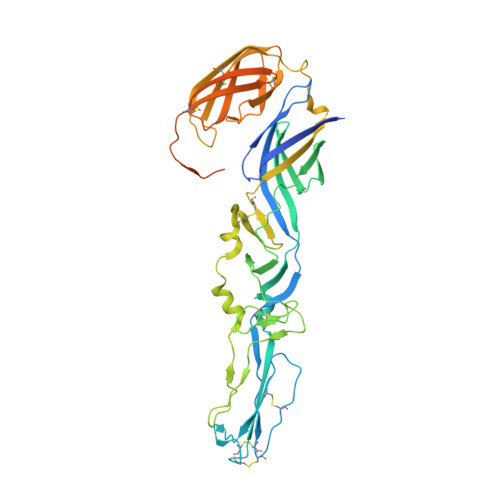
Glycoprotein organization of Chikungunya virus particles revealed by X-ray crystallography.
Voss, J.E., Vaney, M.C., Duquerroy, S., Vonrhein, C., Girard-Blanc, C., Crublet, E., Thompson, A., Bricogne, G., Rey, F.A.(2010) Nature 468: 709-712
- PubMed: 21124458 Search on PubMed
- DOI: https://doi.org/10.1038/nature09555
- Primary Citation Related Structures:
2XFB, 2XFC, 3N40, 3N41, 3N42, 3N43, 3N44 - PubMed Abstract:
Chikungunya virus (CHIKV) is an emerging mosquito-borne alphavirus that has caused widespread outbreaks of debilitating human disease in the past five years. CHIKV invasion of susceptible cells is mediated by two viral glycoproteins, E1 and E2, which carry the main antigenic determinants and form an icosahedral shell at the virion surface. Glycoprotein E2, derived from furin cleavage of the p62 precursor into E3 and E2, is responsible for receptor binding, and E1 for membrane fusion. In the context of a concerted multidisciplinary effort to understand the biology of CHIKV, here we report the crystal structures of the precursor p62-E1 heterodimer and of the mature E3-E2-E1 glycoprotein complexes. The resulting atomic models allow the synthesis of a wealth of genetic, biochemical, immunological and electron microscopy data accumulated over the years on alphaviruses in general. This combination yields a detailed picture of the functional architecture of the 25 MDa alphavirus surface glycoprotein shell. Together with the accompanying report on the structure of the Sindbis virus E2-E1 heterodimer at acidic pH (ref. 3), this work also provides new insight into the acid-triggered conformational change on the virus particle and its inbuilt inhibition mechanism in the immature complex.
- Institut Pasteur, Département de Virologie, Unité de Virologie Structurale, 25 rue du Dr Roux, 75724 Paris Cedex 15, France.
Organizational Affiliation: