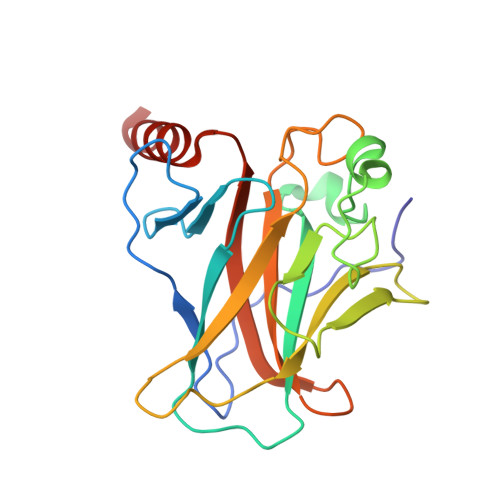
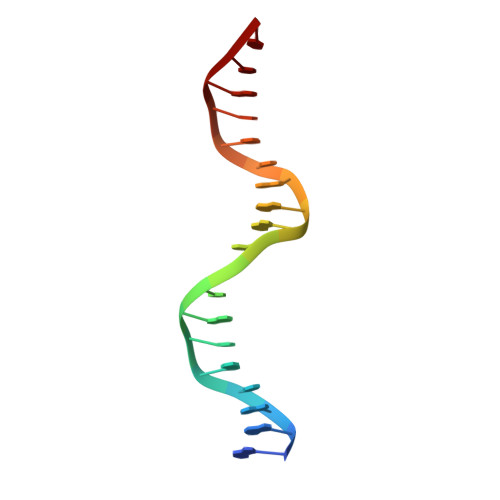
Crystal structure of the p53 core domain bound to a full consensus site as a self-assembled tetramer.
Chen, Y., Dey, R., Chen, L.(2010) Structure 18: 246-256
- PubMed: 20159469 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2009.11.011
- Primary Citation Related Structures:
3KMD - PubMed Abstract:
Recent studies suggest that p53 binds predominantly to consensus sites composed of two decameric half-sites with zero spacing in vivo. Here we report the crystal structure of the p53 core domain bound to a full consensus site as a tetramer at 2.13A resolution. Comparison with previously reported structures of p53 dimer:DNA complexes and a chemically trapped p53 tetramer:DNA complex reveals that DNA binding by the p53 core domain is a cooperative self-assembling process accompanied by structural changes of the p53 dimer and DNA. Each p53 monomer interacts with its two neighboring subunits through two different protein-protein interfaces. The DNA is largely B-form and shows no discernible bend, but the central base-pairs between the two half-sites display a significant slide. The extensive protein-protein and protein-DNA interactions explain the high cooperativity and kinetic stability of p53 binding to contiguous decameric sites and the conservation of such binding-site configuration in vivo.
- Molecular and Computational Biology, Department of Biological Sciences, University of Southern California, Los Angeles, CA 90089, USA.
Organizational Affiliation: