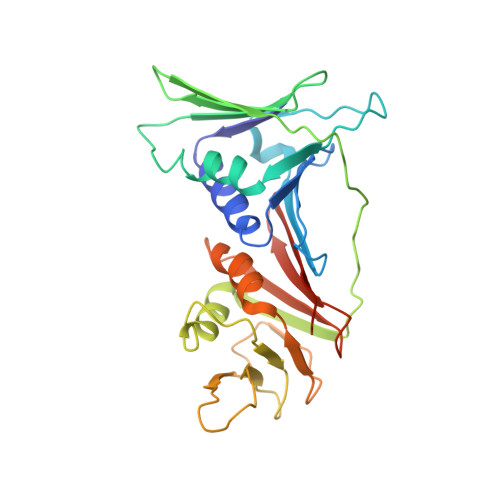
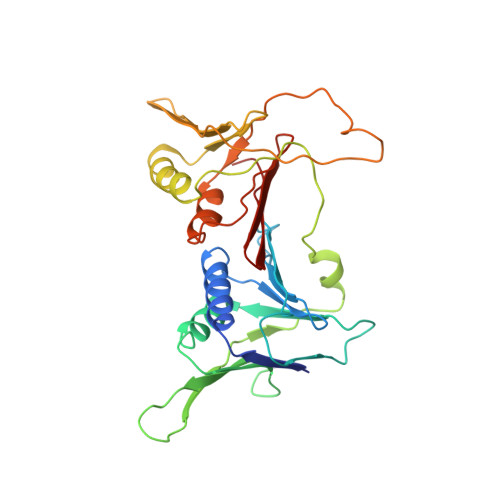
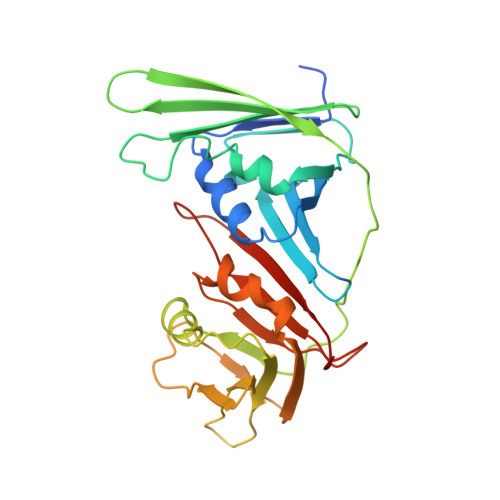
Structure and functional implications of the human rad9-hus1-rad1 cell cycle checkpoint complex
Xu, M., Bai, L., Gong, Y., Xie, W., Hang, H.Y., Jiang, T.(2009) J Biological Chem 284: 20457-20461
- PubMed: 19535328 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.C109.022384
- Primary Citation Related Structures:
3GGR - PubMed Abstract:
Cellular DNA lesions are efficiently countered by DNA repair in conjunction with delays in cell cycle progression. Previous studies have demonstrated that Rad9, Hus1, and Rad1 can form a heterotrimeric complex (the 9-1-1 complex) that plays dual roles in cell cycle checkpoint activation and DNA repair in eukaryotic cells. Although the 9-1-1 complex has been proposed to form a toroidal structure similar to proliferating cell nuclear antigen (PCNA), which plays essential roles in DNA replication and repair, the structural basis by which it performs different functions has not been elucidated. Here we report the crystal structure of the human 9-1-1 complex at 3.2 A resolution. The crystal structure, together with biochemical assays, reveals that the interdomain connecting loops (IDC loop) of hRad9, hHus1, and hRad1 are largely divergent, and further cocrystallization study indicates that a PCNA-interacting box (PIP box)-containing peptide derived from hFen1 binds tightly to the interdomain connecting loop of hRad1, providing the molecular basis for the damage repair-specific activity of the 9-1-1 complex in contrast to PCNA. Furthermore, structural comparison with PCNA reveals other unique structural features of the 9-1-1 complex that are proposed to contribute to DNA damage recognition.
- National Laboratory of Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, 15 Datun Road, Chaoyang District, Beijing 100101.
Organizational Affiliation: