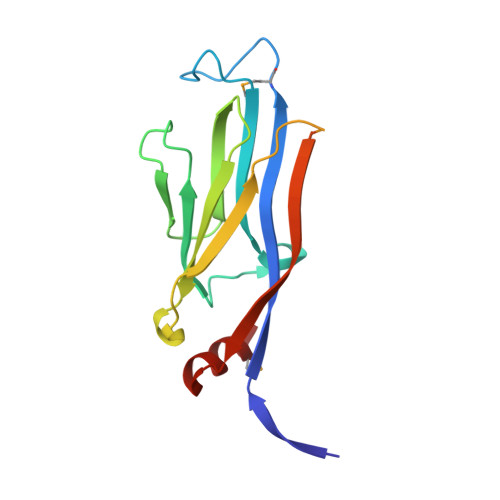
Asf1-like structure of the conserved Yaf9 YEATS domain and role in H2A.Z deposition and acetylation
Wang, A.Y., Schulze, J.M., Skordalakes, E., Gin, J.W., Berger, J.M., Rine, J., Kobor, M.S.(2009) Proc Natl Acad Sci U S A 106: 21573-21578
- PubMed: 19966225 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0906539106
- Primary Citation Related Structures:
3FK3 - PubMed Abstract:
Chromatin can be modified by posttranslational modifications of histones, ATP-dependent remodeling, and incorporation of histone variants. The Saccharomyces cerevisiae protein Yaf9 is a subunit of both the essential histone acetyltransferase complex NuA4 and the ATP-dependent chromatin remodeling complex SWR1-C, which deposits histone variant H2A.Z into euchromatin. Yaf9 contains a YEATS domain, found in proteins associated with multiple chromatin-modifying enzymes and transcription complexes across eukaryotes. Here, we established the conservation of YEATS domain function from yeast to human, and determined the structure of this region from Yaf9 by x-ray crystallography to 2.3 A resolution. The Yaf9 YEATS domain consisted of a beta-sandwich characteristic of the Ig fold and contained three distinct conserved structural features. The structure of the Yaf9 YEATS domain was highly similar to that of the histone chaperone Asf1, a similarity that extended to an ability of Yaf9 to bind histones H3 and H4 in vitro. Using structure-function analysis, we found that the YEATS domain was required for Yaf9 function, histone variant H2A.Z chromatin deposition at specific promoters, and H2A.Z acetylation.
- Centre for Molecular Medicine and Therapeutics, Child and Family Research Institute, and Department of Medical Genetics, University of British Columbia, Vancouver, BC, Canada V5Z 4H4.
Organizational Affiliation: