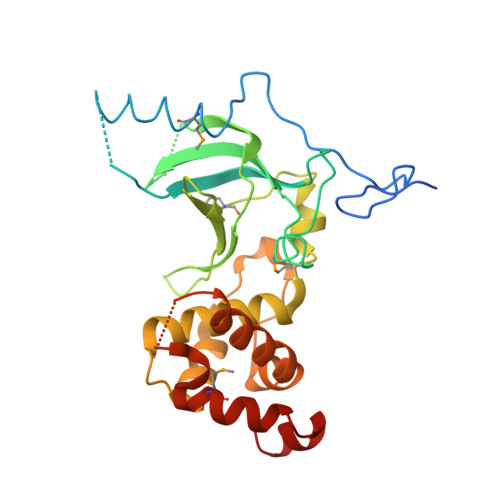
The replication focus targeting sequence (RFTS) domain is a DNA-competitive inhibitor of Dnmt1.
Syeda, F., Fagan, R.L., Wean, M., Avvakumov, G.V., Walker, J.R., Xue, S., Dhe-Paganon, S., Brenner, C.(2011) J Biological Chem 286: 15344-15351
- PubMed: 21389349 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M110.209882
- Primary Citation Related Structures:
3EPZ - PubMed Abstract:
Dnmt1 (DNA methyltransferase 1) is the principal enzyme responsible for maintenance of cytosine methylation at CpG dinucleotides in the mammalian genome. The N-terminal replication focus targeting sequence (RFTS) domain of Dnmt1 has been implicated in subcellular localization, protein association, and catalytic function. However, progress in understanding its function has been limited by the lack of assays for and a structure of this domain. Here, we show that the naked DNA- and polynucleosome-binding activities of Dnmt1 are inhibited by the RFTS domain, which functions by virtue of binding the catalytic domain to the exclusion of DNA. Kinetic analysis with a fluorogenic DNA substrate established the RFTS domain as a 600-fold inhibitor of Dnmt1 enzymatic activity. The crystal structure of the RFTS domain reveals a novel fold and supports a mechanism in which an RFTS-targeted Dnmt1-binding protein, such as Uhrf1, may activate Dnmt1 for DNA binding.
- Structural Genomics Consortium and Department of Physiology, University of Toronto, Toronto, Ontario M5G 1L7, Canada.
Organizational Affiliation: